The structures of most medical devices are far too complex to mold as a single piece. Therefore, it is necessary to assemble their components into a finished product. While this may be done with mechanical fasteners or adhesives, plastic welding may offer significant advantages. This article discusses two important plastic welding technologies: ultrasonic welding and laser welding and their implications for medical device designers and manufacturers.
Ultrasonic Welding: How It Works
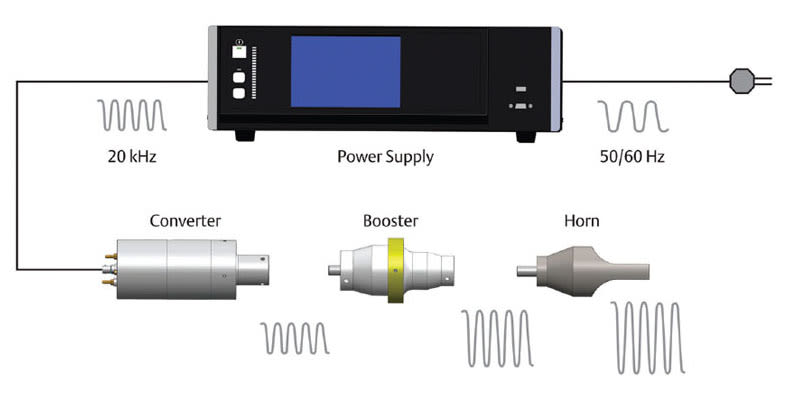
In ultrasonic welding, high-frequency vibrations are applied to two parts or surfaces by a vibrating tool, commonly called a horn or sonotrode. Welding occurs as the result of heat generated at the interface between the parts. The ultrasonic vibrations are created by a series of components — the power supply, converter, booster, and horn — that deliver mechanical vibration to the parts.
As seen in Figure 1, the power supply takes a standard electrical line voltage and converts it to an operating frequency (in this illustration, 20 kHz). This electrical energy is sent through a radio-frequency cable to the converter. The converter uses piezoelectric ceramics to convert the electrical energy to mechanical vibrations at the operating frequency of the power supply. This mechanical vibration is either increased or decreased based on the configuration of the booster and horn. The proper mechanical vibration, known as amplitude, is determined by an applications engineer and is based on the thermoplastic materials used in the parts.
In operation, mechanical vibrations are delivered to the parts to be welded. The parts also are put under a mechanical load, generally with a pneumatic actuator that holds the booster and horn. Under this load, the mechanical vibrations are transmitted to the interface between the material surfaces, which focuses the vibration to create intermolecular and surface friction. This friction creates heat and a subsequent melt, which solidifies into a welded bond.
Ultrasonic welds are widely used to join thermoformed plastic assemblies in medical devices because they eliminate the need for chemical solvents and adhesives, which can raise biocompatibility concerns, and they eliminate the need for mechanical fasteners. The ultrasonic welding process integrates easily into high-volume part production and automation because weld cycles are so fast — typically less than two seconds — and no consumables are required. Minimally invasive surgical instruments, including catheters, cannulas, luers, and trocars, often utilize ultrasonic joining with great success. For example, components that make up the sealing mechanism of a surgical trocar (see Figure 2) are ultrasonically welded into one sub-assembly of the finished product.
Many thermoplastics, both amorphous (such as polystyrene) and semi-crystalline (such as nylon), can be ultrasonically welded. In ideal situations, both parts in a weld are made of the same material. However, dissimilar plastics can also be ultrasonically welded if their melting temperatures (e.g., glass transition temperature, Tg), are similar.

Typical plastic component designs require only minor modification to ensure repeatable, high-strength ultra-sonically welded assembly. The most common (and often “mold safe”) modification adds a small, triangular energy director to one of the mating parts surfaces (see Figure 3) and a corresponding textured surface on the opposing mating surface. The energy director melts and flows to join the two surfaces. Under ideal conditions, polymer chains from each side of the mating parts migrate across the interface and become indistinguishable from the parent material. The result is a bond that is comparable in strength to the parent material.

An Even Cleaner Alternative: Laser Welding
The market for in vitro diagnostics and implantable medical sensors, analyzers, and drug-delivery devices is exploding. But in applications like these, where the vibration required for ultrasonic welding might be undesirable, laser welding is often the best alternative. It provides a cosmetically attractive connection without causing deformation of intricate features or small parts. It also creates hermetic seals between small parts generating minimal flash or particulates, a quality essential to products that require cleanroom-quality assembly and packaging and that are trusted to deliver precise drug delivery therapies.
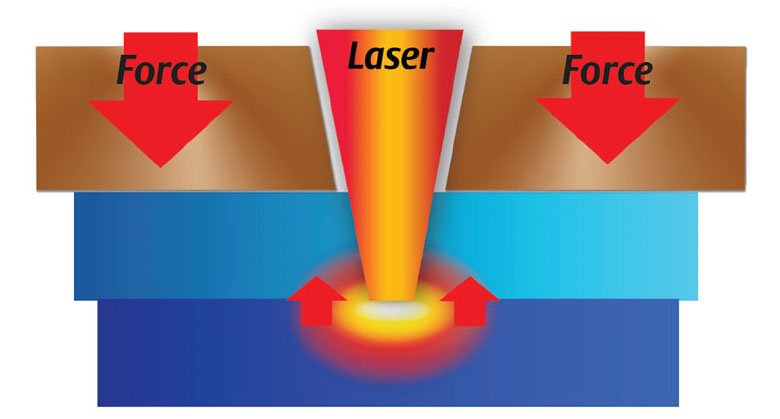
Unlike the ultrasonic method described earlier, laser welding does not introduce mechanical motion between the components being welded. Instead, the near infrared heat needed for part welding is provided by a laser light source. With no relative movement between the parts, weld alignment and part-to-part sealing is exceptionally precise. Welds are fast and clean, with minimal particulate and flash.

A Branson innovation to laser welding, called Simultaneous Through Transmission Infrared welding (STTIr), is the first laser welding technology that can illuminate the entire welding surface at the same time. This approach eliminates the need to move or scan heat sources along the length of the weld and thus dramatically speeds up the laser welding cycle, reducing it to as little as 1–2 seconds on small parts. The exceptional cleanliness and precision of laser welding is the primary factor behind its recent and rapid growth in medical device, consumer electronics, high-end automotive, and business applications.
STTIr laser welding has certain part design requirements. First, one component of the assembly must be transmissive or have low absorbance of the laser wavelength to be used, while the mating component must be absorbent to that wavelength. Second, the geometry of the assembly must allow for delivery of the laser energy to the weld zone, where the melt occurs in the upper section of the absorbent part. There are many ways to bring laser energy to the weld zone. The illustration in Figure 4 demonstrates the fundamental concept.
Laser welding equipment typically consists of a stand that holds one or more laser light sources, or diodes, typically in the 780–980 nm range. The light from these sources is concentrated and delivered to the parts through fiber optic bundles that are arranged around the perimeter of the part to be welded, creating configurable weld zones. This allows laser energy to be increased or decreased within a zone to compensate for part variations or delicate part features. The laser output from each diode is distributed over the weld area of the part according to the heating density required by the application.

Like other types of plastic welding, laser welding utilizes tooling to hold the parts that are to be joined. The key difference in tooling for laser welding is that the fiber bundles must be linked to the tool. Fortunately, this is a straightforward process since the output sections of the fiber bundles are manufactured with circular ferrules or other features that are easy to incorporate into the tool.
Meeting the requirement of having one part that transmits laser light and another that absorbs the light need not be difficult. There are many clear plastic materials, as well as colored pigments, that properly transmit laser light, despite the fact that the parts made from them appear pigmented to the human eye. The same holds true for absorbent parts. Beyond the obvious — carbon black — there are a wide range of color pigments that absorb laser light, resulting in parts that are laser-weldable. Laser-absorbing inks can be pad printed or ultrasonically sprayed onto the weld interface or added to the resin prior to molding. To be sure the combination of part colors and pigments work properly, it is important to consult with the weld equipment supplier.
Figure 5 shows a Branson 3G laser welding system that includes the power supply, controls, and cooling system on the right along with the actuator and laser banks on the left. The system can be mounted on a bench or, in modular form, easily incorporated into automation.
Support for UDI compliance
Unique Device Identification (UDI) programs, which are being phased in in the United States and Europe, will require that, by 2020, UDIs be placed on medical device labels, on packages, and on all medical devices that are designed for reprocessing and multiple use. Beyond device materials and components, a core element of a device UDI is its assembly history.
Fortunately, ultrasonic welders can be equipped to capture, store, and download detailed weld data, assembly data, and audit trails. These data not only simplify compliance with UDI and traceability requirements, but also enhance patient safety. For example, the Branson 2000 XC ultrasonic welder can capture and store weld data for up to 100,000 device assemblies and download it for inclusion in UDI and quality databases (see Figure 6). Its onboard security supports compliance with FDA medical manufacturing regulations, including 21 CFR part 11.
Conclusion
There are many choices, questions, and challenges inherent in the design, development, and production of reliable, repeatable joining process solutions. A material joining supplier provides expertise that can provide valuable guidance on everything from proof of concept to prototype development, scalability, data collection, regulatory compliance, and more.
Involving an experienced engineering expert in early stages can help a manufacturer determine the best joining technology for a particular product. For medical device OEMs that design and manufacture on multiple continents, it is also valuable to partner with a technology supplier with global capabilities that can provide local welding design and production expertise and support wherever device assembly is performed.
This article was written by Tom Hoover, Senior Market Segment Manger, Medical & Business Consumer Electronics, for Branson Ultrasonics Corp., Danbury, CT. For more information, Click Here .



