Briefs: Manufacturing & Prototyping
When bringing new medical equipment or portable mobile medical devices to market, manufacturers turn to packaging engineers to develop custom case solutions that can enhance and...
Applications: Manufacturing & Prototyping
It’s not always easy to walk in someone else’s shoes. It’s even more difficult if those shoes belong to a person with an artificial leg. However, that’s exactly what...
Features: Medical
Imagine you are recovering from an operation and are fitted with wireless body sensors that allow you to move in the hospital bed or around the room in comfort. Once past the...
Features: Medical
Particulate testing of cardiovascular medical devices is an important and valuable step...
INSIDER: Medical
Researchers at the University of Hertfordshire have developed a robotic glove prototype for stroke sufferers. The device can be used in homes to support rehabilitation and personal independence.
INSIDER: IoMT
Medical device regulators at the FDA have issued correcting amendments to their post-market electronic Medical Device Reporting (eMDR) requirements to eliminate any disparities between those rules and their Unique Device Identification (UDI) system.
News: Medical
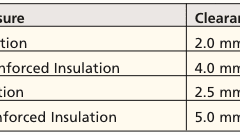
AAMI, the Association for the Advancement of Medical Instrumentation, is leading the small-bore connectors initiative, an international effort to decrease tubing misconnections and increase patient safety. Recently, AAMI has released two additional standards focusing on how to design connectors...
Technology Leaders: Tubing & Extrusion
PVC and polyurethane are thermoplastic materials that can be formed into a variety of shapes, including tubing and reinforced hose. Both materials are useful in numerous applications. But depending on...
Technology Leaders: Medical
With catheters and medical devices becoming increasingly advanced and specialized, so has the process of creating that device. With complex designs, or...
Global Innovations: Energy
Okinawa Institute of Science and Technology Graduate University, Okinawa, Japan
www.oist.jp
Transforming liquids into gels plays an important role in many industries, including cosmetics, medicine,...
Briefs: Medical
New research in robotics to help with stroke rehabilitation, guide wheelchairs, and assist children with Autism Spectrum Disorder are some of the projects now being funded by the National...
Features: Software
Medical device manufacturers operate in a challenging environment filled with stringent regulatory requirements and industry pressures. With a rise in mainstream competitors...
Features: Medical
Not only are medical devices expected to function as intended, they must meet ergonomic, safety, FDA and functional requirements. They must be designed to function in adverse...
Features: Electronics & Computers
Board cleaning is perhaps one of the most overlooked aspects of printed circuit board (PCB) assembly. But savvy medical electronics original equipment manufacturers (OEMs) have a keen sense of...
News: Medical
Health Canada, the Canadian medical device market regulator, is set to require all reprocessed single-use medical devices to Canadian Medical Device Regulations (CMDR) by September 1, 2015. According to MEDEC, the national association created for the Canadian medical technology...
INSIDER: Robotics, Automation & Control
A Ruhr-University Bochum researcher has developed a plasma sterilizer that is specifically suited for ridding medical instruments of germs efficiently, without damaging the material.
INSIDER: Regulations/Standards
The FDA recognizes that the progression to digital health offers the potential for better, more efficient patient care and improved health outcomes, which requires that many medical devices be interoperable with other types of medical devices and with various types of health information technology...
Briefs: Materials
Technipaq, Inc., Crystal Lake, IL
At the onset of a project it seems there are a myriad of things that an engineer or packaging professional should consider when choosing an effective material...
Products: Materials
Bemis Company, Inc., Neenah, WI, has commercially-available alternatives to Barex resin-based packaging. Its new sealant structure, CXB™, is specifically designed for the healthcare market. This sealant is...
Features: Medical
Medical device manufacturers frequently face unique industry challenges, including the need to manage quality processes across disparate sites or...
Features: Medical
The third edition of IEC 60601-1 has been in effect since June 2012. It replaces the previous version as a basic standard for medical electrical equipment and describes the general...
Features: Test & Measurement
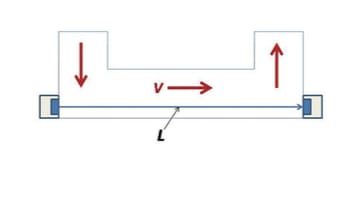
One of the more common purposes of an ultrasonic flow meter is to measure the velocity of a fluid in order to calculate the volumetric flow rate of a medium through a tube. This can be done through the use...
INSIDER: Medical
A group of Korea Advanced Institute of Science and Technology (KAIST) researchers explored how the attractive physical features of zinc oxide (ZnO) materials could be more effectively used to tap into abundant mechanical energy sources to power micro devices.
Mission Accomplished: Sensors/Data Acquisition
In the mid-1990s scientists at NASA Kennedy Space Center were experimenting with an unusual substance: cow digestive bacteria. Could it break down leftover dead plant matter in...
Technology Leaders: Electronics & Computers
While a number of countries have standards in regards to overall medical equipment, a few countries have related component requirements (e.g. plugs...
Global Innovations: Medical
National University of Singapore
www.nus.edu.sg
Regaining mobility after a stroke or other neurological conditions such as spinal cord injuries, traumatic brain injuries, and Parkinson’s disease is often...
Briefs: Sensors/Data Acquisition
Diabetes is the leading cause of limb loss, accounting for more than 65,000 amputations a year nationwide. In addition, there were more than 1,500 major limb amputations from US battle injuries in...
Products: Test & Measurement
TEGAM, Inc., Geneva, OH, has introduced five new Kelvin probes designed for heavy-duty applications or extremely tight spaces, depending on model. When used with TEGAM's R1L bond meters or the 1740 or 1750 microohmeters,...
Products: Software
Teseq, Edison, NJ, has enhanced its NSG 438 electrostatic discharge (ESD) simulator to include a brand new color touch display, allowing changes to settings and features right on the pistol. This is the first ESD...

 Podcasts: Medical
Podcasts: Medical Podcasts: Design
Podcasts: Design Podcasts: Design
Podcasts: Design On-Demand Webinars: Medical
On-Demand Webinars: Medical Podcasts: AR/AI
Podcasts: AR/AI Podcasts: Manufacturing & Prototyping
Podcasts: Manufacturing & Prototyping