After a 20-year effort to establish standards which would minimize the risk of medical misconnections, the pending release of the ISO 80369 series of standards has now reached a point of critical mass, shifting from a development stage to an implementation stage. The new series of standards is destined to change small bore tubing connector designs globally.
The Lure of the Luer
To do or not to do, that becomes the timing question with standards changes. What impact does a change have on your product design decisions and when? The pending release of ISO 80369 raises the question for designers using small bore connectors.
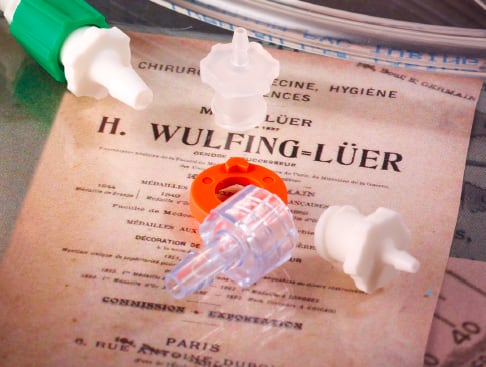
Small bore connectors are components used to connect medical devices to tubing and accessories that deliver fluids and gases to patients. Small bore refers to the size of the opening of the connector (less than 8.5 mm). The most commonly used small bore connectors are luer connectors, also called luer fittings or luers. (See Figure 1)
There are two types of luer connectors: slips and locks. A slip luer consists of a tapered male tip and wider-tapered female port that slide into one another and are held together with simple friction. A luer lock has an additional thread locking sleeves on each half to secure the two halves of the connection together.

Luer connectors present a simple, universal design that provides an acceptable connection for many uses. (See Figure 2) Their widespread use in diverse patient care applications permit connection between unrelated patient delivery systems with different intended uses. Patients might be connected to multiple medical devices. (See Figure 3)
These different devices access different points in the body providing different fluids or treatments:
- Vascular: access into the bloodstream
- Enteral: access into the stomach
- Respiratory: breathing gases for the airway
- Epidural: access into the spine
- Limb Cuff: gases driven to provide pressure to a cuff, such as a blood pressure cuff
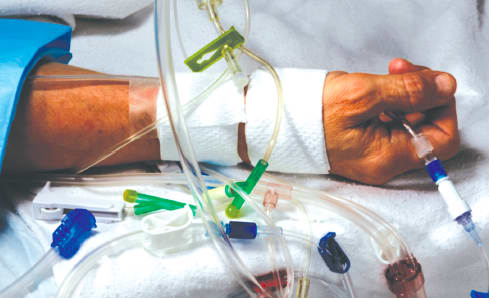
Luer’s universality of application, coupled with the escalation in the number of tubes and connections in medical laboratories, hospital rooms, and operating rooms increases the danger of inadvertent misconnection of wrong systems by health care providers. Such misconnections can cause medication, liquid feeding formulas, or air to be delivered through the wrong tubing to the wrong point in the patient. These errors have resulted in patient injury and death. The U.S. Food and Drug Administration (FDA) has received reports of serious and even fatal misconnections, as has the Institute for Safe Medication Practices (ISMP), The Joint Commission (TJC), the United States Pharmacopeia, and the ECRI Institute. Efforts to mitigate the problem, such as employing color-coded connectors and awareness training, have not proven successful in eliminating the problem.
Developing ISO 80369
To effectively address the issue, small bore connector designs needed to change. However, without a coordinated effort to create dimensionally unique connectors for different applications, preventing misconnection would be difficult to achieve. This initiative to develop a series of unique small bore connectors was first set in motion in Europe in the late 1990s. (See Figure 4)

In 2000, The European Committee for Standardization (CEN) formed a committee to specifically address this issue. The committee created definitions and a framework for the standards by evaluating the different medical applications and products using luers. The committee then reviewed these applications by the risk associated with the potential for a misconnection. This framework is still in use today.
By 2007, an international group of manufacturers, clinicians, and regulators, including the Association for the Advancement of Medical Instrumentation (AAMI) and the FDA, collaborated with the International Organization for Standardization (ISO) to transfer responsibility for this work and move the standard to a truly global effort.
AAMI took a leadership role by organizing Technical Committees and Working Groups to produce the ISO 80369 standards, and recommend practices and technical information reports for medical device compliance to these standards.
The ISO 80369 standard specifies the healthcare fields in which these small bore connectors are intended to be used, divided into seven sections. Part 1 of the standard identifies the general requirements for small bore connectors. The following six sections identify specific requirements for connectors falling into one of the following general application areas:
- Part 2: breathing systems and driving gases;
- Part 3: enteral (feeding tube) and gastric;
- Part 4: urethral and urinary;
- Part 5: limb cuff inflation or non-invasive blood pressure;
- Part 6: neuraxial devices; and
- Part 7: intravascular or hypodermic.
ISO 80369-1 became the first standard to publish in January 2011. It provides general requirements for small bore connectors for liquids and gases in healthcare applications. It also establishes a framework for testing connectors to ensure non-interconnectability of unrelated delivery systems. International standards for non-luer-compatible, delivery system-specific connections have since been under development. Each additional standard in the series (Parts 2 through 7) focuses on connectors for the six specific clinical applications.

The new small bore tubing connector designs are dimensionally driven. The design methodology is based on human-factors engineering and computer-aided design (CAD) analysis to reduce the likelihood of misconnections. The proposed unique designs for the connectors go through a rigorous process to ensure they connect only to the proper mating connector. The standard proposes that the materials used to make these connectors have a modulus of elasticity of 700 kg/cm2 to prevent interconnectability when force fitted. Proposed designs are validated through hands-on usability testing to confirm that they meet their intended purpose of preventing the connection of a medical device from one delivery system to a device of another delivery system. Analysis of design drawings and physical force-fit testing are used to verify that connectors that are supposed to connect do so securely, while connectors that should not join are physically prevented from doing so. (See Figure 5)
The system required to approve the ISO 80369 standards is a methodical and tedious process of coordinating and integrating data and testing from participant manufacturers, clinicians and regulators. These actions take considerable time for review, comment and incorporation into the final standard and involve representatives from around the world.
January 2015 Committee Update on ISO 80369
Recently, ISO 80369 AAMI/ISO working group for small bore connectors met in Melbourne, Australia, from January 20-23, 2015. Each ISO 80369 standard is at various stages of completion. Following is an updated status on each sub-standard’s expected release, per the AAMI website:
- ISO 80369-1: (General Requirements). This standard published in January 2011
- ISO 80369-2: (Breathing Systems). The standard is expected to publish in May 2016.
- ISO 80369-3: (Enteral Feeding). This standard is expected to publish in June 2015.
- ISO 80369-4: (Urinary and Urethral). Currently no work has started on this standard, and no current publish date has been established.
- ISO 80369-5: (Limb Cuff Inflation). This standard is expected to publish in October 2015.
- ISO 80369-6: (Neuraxial Devices). This standard is expected to publish in July 2015.
- ISO 80369-7: (Intravascular or Hypdermic). This standard could publish in October 2015, replacing current ISO594-1 and ISO 594-2 standards for luer fittings. • ISO 80369-20: (Common Test Meth - ods). This standard is expected to publish in May 2015.
FDA Guidance
On February 11, 2015, the FDA published final guidance for small bore connectors used in enteral applications (ISO 80369-3). The agency also recognized standards for small bore connector testing (ISO 80369-20). Following is the FDA’s latest information on this guidance.
This guidance recommends that manufacturers design and test enteral connectors based upon AAMI/CN3:2014 (PS), Small bore connectors for liquids and gases in healthcare applications – Part 3: Connectors for enteral applications, and AAMI/CN20:2014 (PS), Small bore connectors for liquids and gases in healthcare applications – Part 20: Common test methods” to ensure that each proposed enteral connector is physically incompatible with non-enteral devices.
For enteral connectors that do not meet AAMI/CN3:2014 (PS), also known as proprietary connectors or transition connectors, this guidance continues to recommend that manufacturers design and test the devices based upon AAMI/American National Standards Institute (ANSI)/ISO 80369-1 standard, Small bore connectors for liquids and gases in healthcare applications – Part 1: General requirements.
The FDA anticipates recognizing future standards for other small bore connector applications as they are developed.
Additionally, this guidance document serves to notify manufacturers submitting 510(k)s or modifying a device already cleared for enteral connectors that color-coding, labeling, and tagging attached to the device, by themselves, are no longer sufficient to satisfy safety concerns regarding misconnection hazards associated with connectors, such as 6° tapered luer connectors. These methods have proven ineffective in eliminating the issue.
Lastly, this guidance recommends that when submitting a 510(k) or modifying an existing product already cleared via 510(k), a manufacturer should perform a risk assessment to assess the risks of the proposed enteral connector misconnecting to non-enteral devices.
FDA’s guidance documents, including this guidance, do not establish legally enforceable responsibilities. Instead, guidances describe the FDA’s current thinking on a topic and should be viewed only as recommendations, unless specific regulatory or statutory requirements are cited.
Compliance
Effective January 11, 2016, a California law (HB 1867) will prohibit general acute care, acute psychiatric, and special hospitals from using an epidural, intravenous, or enteral feeding connector that fits into a connection port other than the type for which it was intended. AAMI expects that all medical device manufacturers and suppliers will comply with the new California law, and develop modified products that incorporate the new connectors and phase out products with old connectors.
The European Union (EU), and other international regulatory bodies, have their own requirements for acceptance of the standards. Foreign medical device manufacturers wishing to sell product into US markets, however, would need to comply with the ISO 80369 standards as per the FDA requirements.
There will be a phase-in period for product development and implementation guided by the FDA, and existing state legislation (for example, California). The standard generally allows three years to manufacturers for new product adoption of the new connector standards, and five years for adoption into existing devices. However, these timelines are influenced by state legislation in the case of enteral and epidural connectors. Market expectations will ultimately determine the speed of conversion from old to new connector.
Shift to Implementation
After a 15-year effort to establish standards which would minimize the risk of medical misconnections, the pending release of the ISO 80369 series of standards has now reached a point of critical mass, shifting from a development stage to an implementation stage.
At this juncture, medical device OEMs should proactively look into how these standards impact their products, and begin developing plans to prepare for the changes. Just six months ago, this necessity may have been premature, and completion of the 15-year effort was still in question. However, it is now clear we are at the beginning of a significant change in small bore connectors that impacts both medical device OEMs and the patients that use their products.
This article was written by Jim Brown, Medical Business Unit Manager, CPC (Colder Products Company), St. Paul, MN. CPC is a member of the Association for the Advancement of Medical Instrumentation (AAMI), and the Bio-Process Systems Alliance (BPSA). The company participates in the ISO 80369 Series of Small Bore Connector Standards working group, and has been actively involved in the development of the entire standard. For more information, Click Here . MD&M East, Booth 1457



