From the Editor: Regulations/Standards
According to the late, great David Bowie, “the stars look very different today”. After two years of collecting the 2.3% Medical Device Excise Tax, the tax has now been suspended for all of 2016 and 2017 when President Obama signed the Consolidated Appropriations Act of 2016. The tax was expected to raise almost $30 billion over...
Briefs: Robotics, Automation & Control
While trips and stumbles leading to falls can be common for amputees using leg prosthetics, a new robotic leg prosthesis being developed at Carnegie Mellon University promises to help users recover their...
INSIDER: Medical
Cybersecurity threats to medical devices are a growing concern that present a potential risk to the safety and effectiveness of medical devices. To address this, the FDA has issued...
INSIDER: Regulations/Standards
In a draft guidance for industry and staff issued on December 31, the FDA proposed notifying the public about medical device “emerging signals that the agency is monitoring or analyzing,...
Products: Medical
Advanced Orientation Systems, Inc., Linden, NJ, has released the wireless EZ-LEVEL-PRO dual axis inclinometer suitable for laboratory, QC, or high-resolution production applications and allows users to perform...
INSIDER: Regulations/Standards
The FDA’s Center for Devices and Radiological Health (CDRH) announced the 2015 Experiential Learning Program (ELP) General Training Program, which is intended to educate CDRH staff regarding the policies, laboratory practices, and challenges faced in broader disciplines that impact the medical...
R&D: Medical
Engineers at the University of Texas at Arlington Research Institute (Fort Worth, TX) and The University of Washington (Seattle, WA) have developed a healing glove that delivers needed medicine to an injured hand. The device, known as the Bioengineered Smart-Glove for Regenerative Healing of...
Products: Medical
Microsemi Corporation, Aliso Viejo, CA, announces the availability of the smallest radio module it has ever produced. The ZL70323 is optimized for implantable medical devices...
INSIDER: Electronics & Computers
IEEE, Piscataway, NJ, has announced a new standard and two new standards development projects designed to support plug-and-play, interoperable communications across eHealth devices. The new eHealth standard is IEEE 2410™-2015, Biometrics Open Protocol Standard,...
Technology Leaders: Medical
The results of recent surveys suggest that more than half of all healthcare decisions are made using test results obtained from medical diagnostic instrumentation. These tests are...
From the Editor: Regulations/Standards
Crowdfunding is being used every day to raise money for various causes and to aid in the launch of new products. Spurred on by themes such as “Be the change you want to see in the world,” donors feel virtuous for helping to fund a child’s medical bills or helping a non-profit to provide clean...
Briefs: Medical
When DARPA launched the Revolutionizing Prosthetics program in 2006, the state of upper-limb prosthetic technology was far behind lower-limb technology. Advancing upper-limb technology was judged...
INSIDER: Medical
On October 14, the FDA issued a draft guidance document to assist industry in designing evaluation strategies for, and reporting the results of, animal studies for medical devices. The studies utilized for the assessment of these devices typically provide initial evidence of device...
INSIDER: Robotics, Automation & Control
VeloX, a prosthetic heart valve developed by National University of Singapore researchers, can be implanted through a small incision for the treatment of a serious heart valve disorder called mitral regurgitation. The device is particularly beneficial to patients who are of high surgical risk or are...
Technology Leaders: Manufacturing & Prototyping
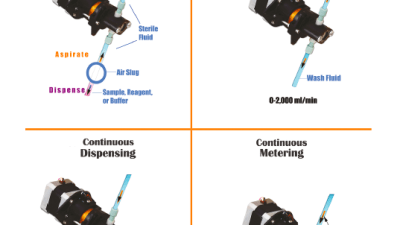
This article addresses how implantable devices can be de - signed to modulate drug infusion safely and...
Technology Leaders: Connectivity
An exciting trend in drug delivery is underway: the movement toward smaller, smarter, wirelessly connected electronic devices that allow patient-administered...
From the Editor: Wearables
Recently, an online discussion board broached the subject of FDA approval for a diagnostic app that might be able to predict when a patient’s condition would relapse. The question postulated whether there was a way to circumvent FDA approval by launching the app in another country and making it available online. And, if...
INSIDER: Medical
While historically, the development of new technologies to improve patients’ lives has relied upon experts’ opinions rather than asking patients and families directly what they consider important, this may be about to change. Patients and their care partners are becoming intimately...
INSIDER: Medical
A team of Johns Hopkins University scientists discovered that noninvasive brain stimulation temporarily improves motor symptoms in patients with Parkinson's disease (PD). The Johns Hopkins study also revealed that people with Parkinson's disease are still capable of making quick,...
INSIDER: Medical
OpenFDA is releasing a treasure trove of information on medical devices that could help spur innovation and advance scientific research. OpenFDA’s Application Programming Interface (API) expands the previous resources about medical device-related adverse events and recalls by incorporating information...
Briefs: Medical
In September 2013, the FDA announced new regulations for medical device manufacturers known as UDI (Unique Device Identifier) that would require all medical devices to bear a label...
Briefs: Medical
Ensuring that device packaging meets specifications.
Considering the complex science and research that goes into developing medical devices, it is important...
INSIDER: Medical
The FDA is seeking companies to take part in a pilot program that would allow medical device manufacturers to report malfunctions of certain low- and medium-risk devices on a quarterly basis. This pilot program will help the agency develop criteria for quarterly malfunction reporting...
INSIDER: Medical
The Emergo Group recently posted an advisory that the FDA has revised the fee charged for issuing medical device export certificates in response to higher costs and demand for these documents.
Technology Leaders: Regulations/Standards
Collaboration among healthcare technology stakeholders—from device manufacturers and healthcare delivery organizations to healthcare security intelligence...
Technology Leaders: Medical
Smart, connected devices are an increasing reality of daily life. It is estimated that by 2020, there will be 33 billion internet devices in the world—enough for 4...
Features: Medical
Most orthopedic implant manufacturers still rely heavily on traditional coatings for their implants, such as sintered bead and plasma-sprayed metallic and hydroxyapatite (HA) coatings. These...
From the Editor: Regulations/Standards
In mid-July, the House of Representatives passed HR 6, also known as the 21st Century Cures Act, which expedites research and development on debilitating diseases and makes it easier to get important treatments to the patients who need them. Among other things, it makes research collaborations easier, reforms and streamlines...
Briefs: Materials
Gold coating could reduce scarring.
A team of scientists from Lawrence Livermore National Laboratory, Livermore, CA, working with other researchers at the University of California, Davis, say that...

 Podcasts: Robotics, Automation & Control
Podcasts: Robotics, Automation & Control Podcasts: Robotics, Automation & Control
Podcasts: Robotics, Automation & Control Podcasts: Design
Podcasts: Design Podcasts: Medical
Podcasts: Medical Podcasts: Medical
Podcasts: Medical On-Demand Webinars: Medical
On-Demand Webinars: Medical