Pump systems are ubiquitous in medical and life science products, from blood pressure monitors and drug-delivery devices, to pipettors and diagnostic instruments. As the demand for smaller, less intrusive — sometimes even wearable — products grow, engineers must meet these expectations without compromising on pump system performance.
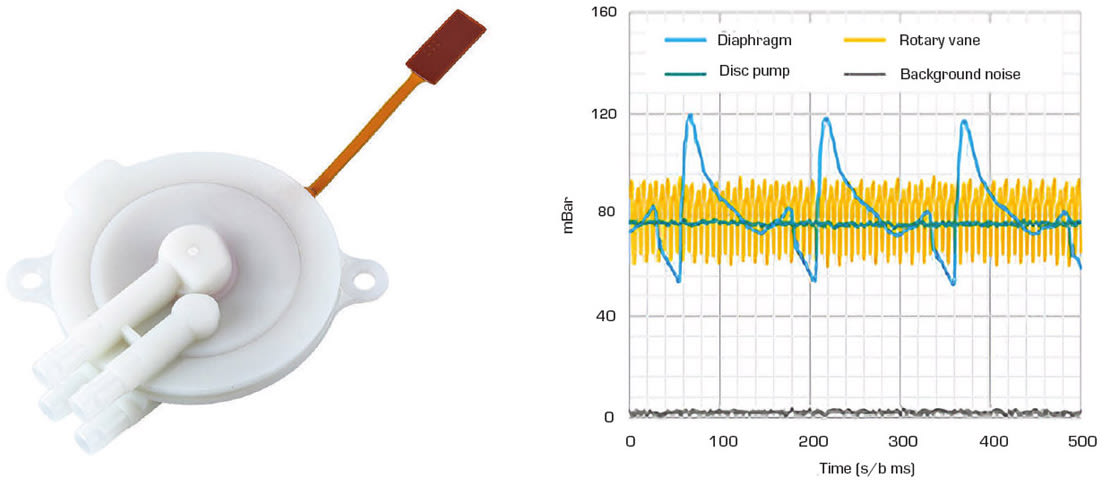
For many years, motor-driven pump technology has been the only solution to this challenge. However, this technology can cause an array of issues that impact the performance of the device and the end user experience. By their design, these pumps are noisy and heavy, suffer from flow pulsation and mechanical vibration, and offer very poor control due to their high inertia and inability to react quickly to set point changes.
When selecting the right pump technology for a device, there used to be a trade-off between operational advantages that improve user experience (small size, low noise, etc.) and pump system performance (flow rate, pressure/vacuum capacity etc.). Often, the higher the capacity of the pump, the heavier and noisier it is.
However, more recently engineers have been turning to a piezo technology known as the disc pump (see Figure 1 and the sidebar, “What Is a Disc Pump”). This technology has a unique set of features including silent operation, compact form factor, pulsation-free operation, zero vibration, and high-precision control. These attributes have helped the disc pump drive innovation across a broad spectrum of medical applications such as point-of-care (POC) diagnostics, wearable intermittent pneumatic compression (IPC), and sleep apnea therapy.
What Is a Disc Pump?
A disc pump is an innovative piezoelectric micropump that takes advantage of nonlinear acoustics and an ultra-fast valve technology to deliver the following features and benefits:
Silent operation
Small size and light weight <5 g
Pulsation-free output
No mechanical vibration
Ultrafast (ms) response to set point changes
Near infinite turndown ratio
This article takes a closer look at two fields in which the disc pump has unlocked innovation: ambulatory blood pressure monitoring and liquid handling.
Ambulatory Blood Pressure Monitoring
While 24-hour ambulatory blood pressure monitoring (ABPM) observation is routine, there are several shortcomings with the current generation of blood pressure monitoring systems. The root of many of these shortcomings is in the use of traditional motor-driven diaphragm pumps. In principle, blood pressure measurement can be taken during cuff inflation. However, airflow pulsation introduced by traditional pump technologies masks the oscillometric signal from the patient, making it difficult to take measurements while the pump is running. Instead, most conventional systems take measurements during cuff deflation once the pump is switched off.
The disc pump, however, has no pulsation, enabling blood pressure readings to be carried out during the cuff inflation cycle. This makes the user experience more comfortable, because the cuff needs only be inflated to the maximum (systolic) blood pressure reading and not much higher — as is the case with the conventional approach. Patient comfort is further enhanced by the pump’s silent, vibration-free operation, which enables measurements to be taken at night with minimal disruption.
In conventional ambulatory blood pressure monitors, the pump tends to be too big and heavy to integrate with the arm-worn cuff. Instead, the module is worn around the neck or on a belt loop and connected to the cuff by a hose. This hose can kink and interfere with measurements, so removing it is an advantage. The disc pump is small and lightweight enough to be directly integrated into arm-worn modules, eliminating the need for a hose.
Pressure Driven Flow/Air-Over Liquid
Pressure driven flow (PDF), also known as air over liquid, is a liquid transport mechanism in which air pressure is used to move liquid through a system. This approach is used in many applications from point-of-care diagnostics to organ-on-a-chip and drug-delivery systems. It addresses several drawbacks of traditional liquid pumping, such as the risk of cross contamination, because the pump never comes into contact with the liquid being handled.
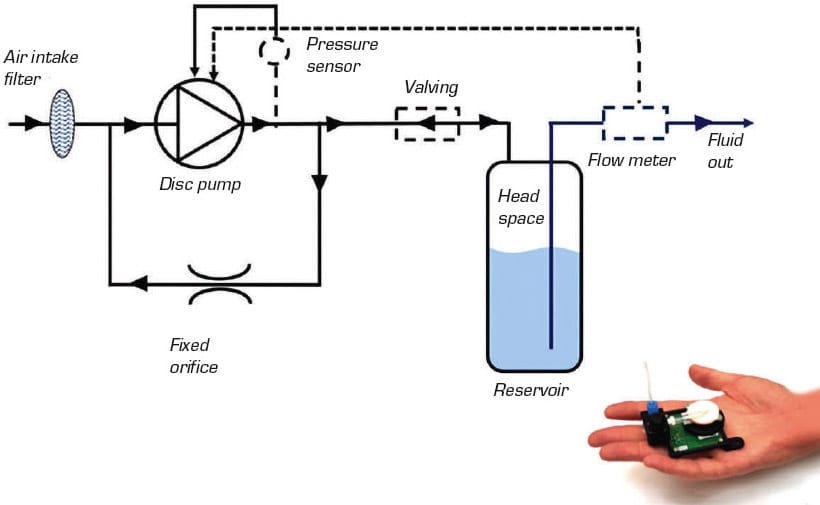
The disc pump is an excellent pressure source for PDF. Operating in a closed-loop system, its high-precision pressure control provides exceptional control over liquid flow. The pulsation-free output and rapid response speed enables liquids to be moved exactly as intended, whether in smooth laminar flow streams for microfluidic research, or oscillating profiles intended to mimic blood flow in the body (see Figure 2).

Using gas pressure to move liquids provides many other benefits over traditional technologies, such as peristaltic pumps and syringe pumps, as detailed in Table 1. As the disc pump can generate both positive and negative pressure, it can also be used for applications like pipetting and time-metered dosing, where the disc pump’s ability to accurately control pressure translates across into accurate liquid control. For example, in time-metered dosing, the head pressure above a body of liquid is accurately controlled, and the dispensed volume is then governed by a fast-acting valve.
Testing in A Project: Disc Pump Development Kit
A development kit includes the electronics and accessories needed to start developing a medical device or fluid handling system. The development kit can drive multiple disc pumps, smart pump modules, and solenoid valves. These products are provided individually, enabling designers to tailor the kit with the solenoid valve and pump model that best fits specific requirements.
This article was written by Emma Wilson, Marketing Manager for Lee Ventus, Cambridge, UK. For more information, contact



