Method for Treating Cartilage Defects
Michael Lytinas
Ideas Foundation, Boston, MA
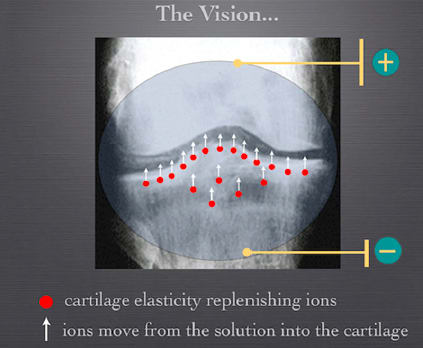
This invention is a method to treat cartilage defects in osteoarthritis and cartilage injuries by moving ions into a collagen matrix of the defective cartilage using electricity. (See Figure 1)
Human joint surfaces are covered by articular cartilage, a low friction, durable material that distributes mechanical forces and protects the underlying bone. Injuries to articular cartilage are common, especially in the knee. Data from the U.S. Centers for Disease Control and Prevention (CDC) and clinical studies have suggested that approximately 100,000 articular cartilage injuries occur per year in the US. Since damaged articular cartilage does not heal, degeneration of the surrounding uninjured cartilage occurs over time. The current osteoarthritis therapeutic regiment includes drugs for pain alleviation, injections of hyaluronic acid or other lubricant-type substances inside the joint to ease the function of the joint, and finally surgery and joint replacement.
This technology differs from what is currently available in the market since it works to bring cartilage back to its previous, healthy stage by forcing ions back into the cartilage. The elasticity that the articular cartilage offers is due to the osmotic swelling of the chondroitin sulphate chains within the cartilage. These chains attract counter ions Na+ to maintain electroneutrality and this creates the osmotic swelling which translates into elasticity of the cartilage in humans. In osteoarthritis, these ions are lost. With this technology, we try to put them back into their place to replenish the cartilage and restore its elasticity and the normal function of the joint.
The clinical part of this technology includes a probe that contains a solution with free ions and touches the defective cartilage part. By applying electricity, the ions move from the solution into the cartilage replenishing it with fresh ions and restoring its function.
For more information, visit http://contest.techbriefs.com/cartilage .
Portable and Automated Sickle Cell Disease Diagnosis and Patient Monitoring Using Magnetic Levitation
Stephanie Knowlton and Dr. Savas Tasoglu
University of Connecticut, Storrs, CT
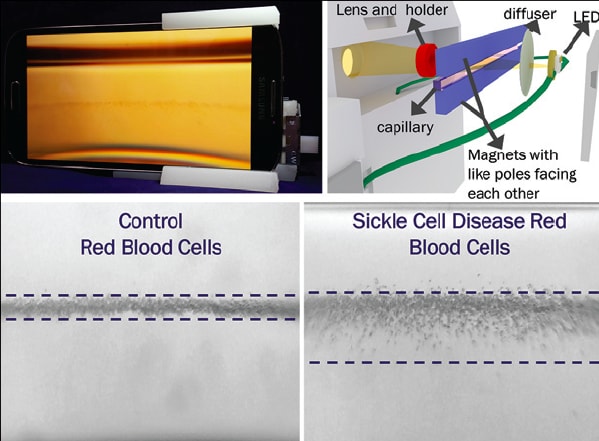
About 300,000 children are born with sickle cell anemia every year and, if left undiagnosed, can lead to “silent” strokes and other serious complications. This hereditary disease is most common in Central and West Africa but in these developing areas, there is no widespread screening program and the mortality rate is 50% to 90% before the age of 5. This portable, economical, and user-friendly device can be used to diagnose sickle cell disease in low-resource settings with no external energy source or equip ment or specific labels or antibodies.
This approach is based on magnetic levitation, which en ables real-time interrogation and monitoring of the biological activity of red blood cells based on subtle changes in their magnetic signature and density. The test requires only a drop of blood obtained via finger stick, which is mixed with a cell-friendly solution containing paramagnetic ions and sodium metabisulfite to deoxygenate and dehydrate sickle cells. Sample preparation takes less than a minute and results are available within 15 minutes. Images of levitating cells are captured using a smartphone camera through an optical lens and analyzed by an application to identify sickle cells, which have a higher density and levitate at a lower height than healthy cells. (See Figure 1)
Current testing procedures cannot differentiate between a patient who has inherited two sickle cell genes (who is said to have sickle cell anemia) and a patient with one sickle cell gene (sickle cell trait). Hemoglobin electrophoresis to distinguish between these cases requires equipment often unavailable in developing countries. Because this platform is based on digital analysis, it can distinguish between sickle cell anemia and trait.
Currently, the devices are 3D printed for rapid prototyping but they may be manufactured at a low cost (estimated under $10) for in low-resource countries and patients living with sickle cell disease. Each test costs around $1, which is only a fraction of the current test cost. Keeping the design simple, said Knowlton, was actually more difficult than making it more complicated, but it was worth it since the current prototype only requires an LED, 2 magnets, a lens, and a smartphone.
For more information, visit http://contest.techbriefs.com/levitation .



