Building on the success of smartphones, mobile gadgets, and wearable sports fitness gear, the medical industry has steadily trended toward improved portability of monitoring devices used in ambulatory patient care. In addition to boasting smaller footprints, many newer medical devices incorporate capacitive and mechanical touch pads, vibrant LED displays, and other technological advances first pioneered in consumer electronics. Worn continuously during normal activities, ambulatory monitoring devices make it possible for many patients requiring continuous monitoring of vital signs to leave the confines of a hospital or other clinical setting.
Thin, Flexible, Lightweight, Durable

A myriad of inherent safety issues are associated with the design decisions made on a device intended to monitor critical vital signs or deliver lifesaving medication. Sensors and cable assemblies used in ambulatory patient monitoring equipment are required to meet FDA and ISO safety and bio-compatibility requirements. In addition to compliance with regulations and power and signal requirements, ambulatory medical devices demand a higher level of mechanical performance to help ensure reliable operation and patient safety.
Portable consumer devices typically come equipped with thin, flexible cable assemblies for power and signal connectivity. While appropriate for a user’s mobile telecommunications or infotainment purposes, the higher performance, safety, and regulatory requirements of portable and ambulatory medical devices prevent the use of the same connectors and cable assemblies used in consumer products.
As the size and bulk of cables used for medical devices decrease, the design must be carefully considered to prevent any loss of mechanical or electrical performance. The cable assembly must still be durable and deliver a service life consistent with the medical device to which it will be attached, while still meeting all safety and regulatory requirements. The right technologies make it possible to design a thin, flexible, lightweight medical device cable assembly with optimal mechanical performance and rugged construction. (See Figure 1)
Proliferation of Interconnect Cable Assemblies
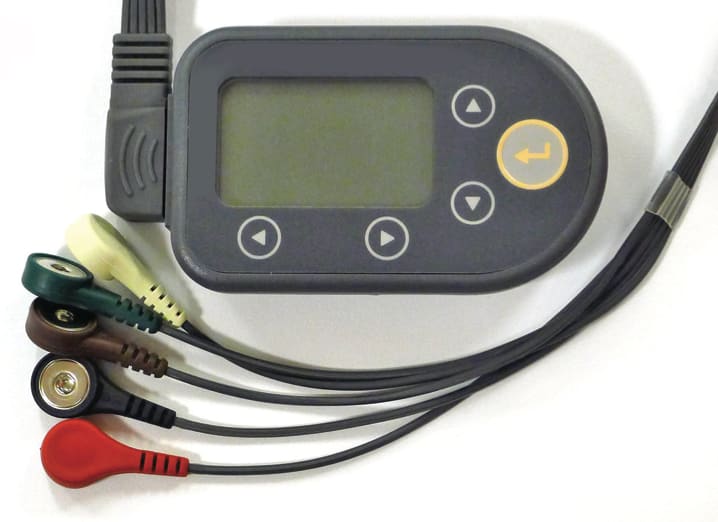
There may come a day when patients won’t require cables to continuously record various vital signs. A number of wireless medical monitoring devices have been successfully released. Yet, even as the term “wireless” becomes more prevalent in consumer and medical markets, the number of device cables continues to increase because of the rising demand for monitoring additional parameters — in conjunction with designs that afford improved patient comfort.
Due to the miniaturization of electronics, portable devices can now perform both patient monitoring and diagnostic ECGs while the patient is fully mobile. Devices the size of a mobile phone can now do the type of monitoring that was once carried out by large stationery monitors commonly found in a hospital setting. (See Figure 2)
Cable design and performance reflect on the quality and performance of the monitoring device. Tremendous design and manufacturing resources are directed toward developing new monitoring technologies that will help people stay healthy and active. Those sophisticated devices can be rendered useless by a poorly designed or low quality cable assembly that results in intermittent operation or equipment failure. When engineering cables for medical monitoring devices, a number of factors must be considered to protect the patient and equipment.
Form + Function Inform Design

The most common way to achieve isolation of the defibrillation pulse is to incorporate a resistor for each ECG lead. The closer the resistor is placed to the patient contact point, the smaller in size the resistor can be, which is more significant for ambulatory devices. A common practice is to incorporate the resistor into the ECG snap or pinch clip by over-molding. (See Figure 3)
Enhancing Service Life and Performance by Strain Relief Design
The ambulatory medical device market benefits from a convergence of technologies that have been well-honed in mobile electronics and, more recently, wearable consumer devices. Wearable electronics for consumers are typically secondary devices, worn occasionally and often connected wirelessly to a smartphone or automotive Bluetooth®.
The importance of size, weight, flexibility, and durability are heightened when a device must be worn by a patient 24/7. The accompanying power and signal connectors and cables will be used in ways that would seldom arise in a clinical setting. They must be able to withstand all activities of daily living, including getting snagged on clothing, tugged, bent, twisted, and otherwise abused.
Strain relief, also referred to as bend relief, is important to the electrical and mechanical integrity and overall performance of a medical cable assembly, especially a cable used for ambulatory patients. Strain relief provides a transition from the flexible cable to a rigid connector or connection point. A properly designed strain relief will prevent mechanical force applied to the exterior of a cable from being transferred to the electrical terminations within the connector or device, which could potentially lead to failure. Permanently attached cables may pass through an opening into the device and be anchored by a grommet, which provides protection against damage from an axially applied load. Strain reliefs can be either premanufactured or custom designed for a given application.
A custom strain relief can be molded over and onto the cable, or designed as a separate unit and slipped onto the cable. Pre-manufactured strain reliefs are slipped over cable before a connector is terminated. Most off-the-shelf connectors offer optional corresponding pre-manufactured strain reliefs that may be available in different colors, which can make device connections more intuitive and user-friendly.
A custom designed and over-molded strain relief typically offers superior flex life, tensile strength, and moisture protection than pre-manufactured strain reliefs. Moisture protection is often an important consideration for ambulatory medical interconnect systems. Off-the-shelf connectors typically incorporate shells and pre-molded boots or collars. When these components are assembled into a finished connector, they often do not offer the required degree of ingress protection. In contrast, custom overmolded connectors can be designed and manufactured with the desired ingress protection rating.
Make No Compromise on Safety or Reliability
Medical device manufacturers are following the lead of consumer electronics to shrink the size of their devices while incorporating even more extensive and sophisticated capabilities. But, as medical devices get smaller, the need for reliability and safety does not diminish. Whereas the failure of a consumer device may be bothersome, the failure of a medical device may compromise patient safety.
Smaller medical devices that require a connection to the patient present unique design and manufacturing challenges. Designing a high-performance cable assembly that is robust and has a long service-life becomes more difficult as the physical size of materials is reduced. Smaller connectors and smaller gauge wire have less inherent strength than larger forms of the same components. To meet these challenges, medical cable and interconnect engineers are creating unique designs and employing new materials and manufacturing processes that lend themselves to smaller sizes.
These and other exciting ideas are surfacing in healthcare device design, aimed at allowing patients greater mobility and allowing clinicians to more effectively and economically monitor and manage the health status of their patients.
This article was written by Hank Mancini, Marketing Manager for Affinity Medical Technologies, Costa Mesa, CA, a subsidiary of Molex Incorporated. For more information, Click Here .



