Until recently, developers and manufacturers of medical devices have not been required to consider security in their products. New guidance from the U.S. Food and Drug Administration (FDA) and expanded European Union requirements for personal data protection now make security design in medical devices a necessity. While IT network attacks get most of the press, it is important to remember that physical attacks, such as accessing a maintenance serial port, can be just as dangerous.
Medical Security in the News

Security experts have reported weaknesses in hospital and clinic networks for several years. Even though these networks contain extremely sensitive patient data and connect life critical equipment, they continue to be proven easy to infiltrate. While networking equipment, like routers, may be state of the art in terms of security, the medical equipment on the network often has little to no security protection. Breaching a device with malware can open a back door to allow remote hackers access to sensitive data across the network, and moreover, cause the device to operate in a dangerous manner.
Since 2015, there has been much regulatory activity on security that affects medical device design. In the EU, the recent adoption of the new General Device Protection Regulation, which applies to all devices, has strict requirements for the protection of personal data. Additional regulations are soon to be released specifically for medical and intravenous delivery devices.
For device designers, the more specific recommendations from FDA provide more useful guidance as to how to meet security requirements. FDA has issued formal guidance on both premarket submissions and postmarket management of security in medical devices. A key item in the premarket guidance states that security hazards should be part of the risk analysis, while the postmarket guidance clearly refers to the need for secure software update procedures. The new postmarket guidance states that FDA typically will not need to clear or approve medical device software changes that are made solely for updating cybersecurity features in the field. This latitude is to enable fast response to emerging threats. Going even further, FDA issued a Safety Communication that was triggered, for the first time, by cybersecurity vulnerabilities of one type of infusion pump. This communication recommended discontinuing the use of several previously approved devices solely based on their vulnerability to attack.
What to Do to Ensure Cybersecurity
While medical equipment developers are experienced at developing systems to meet functional safety requirements, cybersecurity adds another dimension to the design process. It is advisable to consult with experts to evaluate the different trade-offs to achieve an appropriate security level for the product. For example, INTEGRITY Security Services (ISS), a Green Hills Software company, helps clients address FDA and EU requirements with an end-to-end embedded security design. ISS supports medical device developers in the application of the following five rules of embedded security.
Rule 1: Communicate without trusting the network. An increasing percentage of medical devices are always connected, and many devices are required to be connected for maintenance or upgrades (see Figure 1). While protecting patient data is critical, a device’s fundamental operating parameters, such as maximum dose limits on infusion pumps, are also critical. Even without sensitive data, if connected on a hospital network, a device can be a target for hackers to penetrate the network.
To prevent a security breach, it is necessary to authenticate all endpoints, including the device itself, any human users interacting with the device, and any other connected systems. Secure designs should never assume users and control software are valid just because the received messages have the correct format. In the infusion pumps that were the target of FDA’s safety communication, a hacker was able to gain access to the network, reverse engineer the protocol, and send properly formed commands that would have allowed a lethal dose of medication to be administered to the patient. Authentication protects medical devices from executing commands originating from unknown sources.
Rule 2: Ensure that software is not tampered with. There are many ways for malware to be injected into a device, including:
- Using a hardware debug interface such as JTAG.
- Accessing test and debug interfaces, such as Telnet and FTP.
- Exploiting control protocols that were developed without considering security.
- Simulating a software update that assumes trustworthiness without verification.
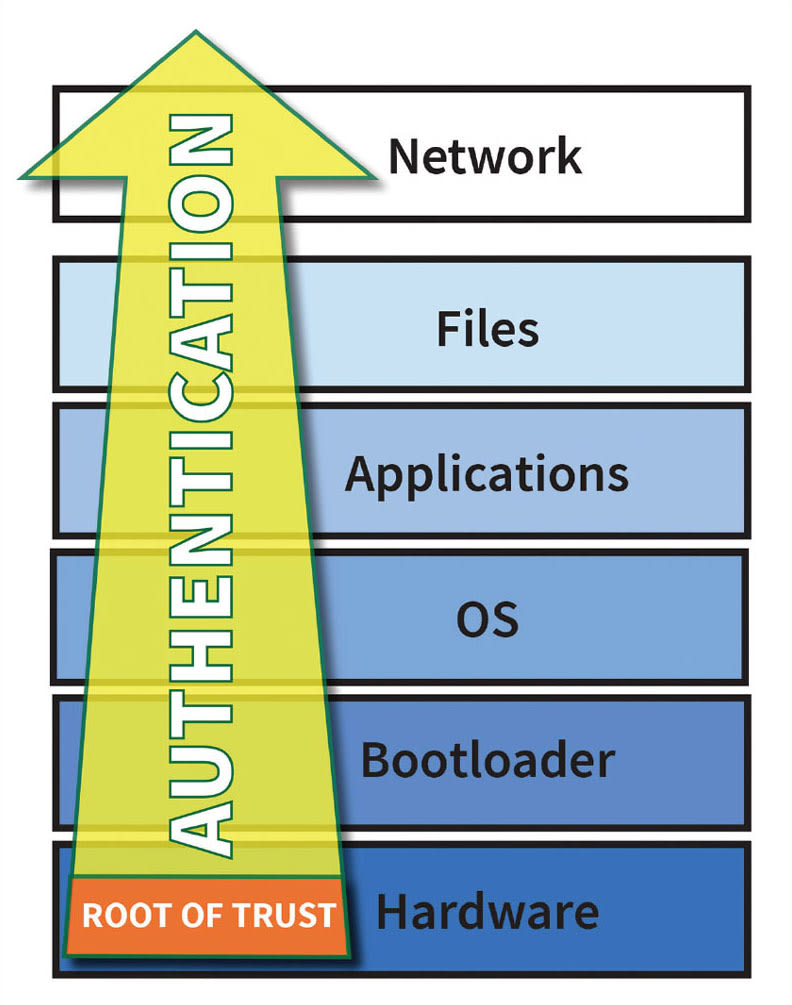
System software should not be trusted until proven trustworthy. The point where authentication starts is called the root-of-trust, and for high-assurance systems, this point must be in either hardware or in immutable memory. A secure boot process starts at the root-of-trust and verifies the authenticity of each software layer before allowing it to execute (see Figure 2).
Secure boot verifies the source and integrity of software using digital signatures. Software is signed during release and verified each time it is loaded. This guarantees that a device is free of malware and operates to the quality it was developed. As a result, by preventing malware, secure boot prevents any targeting of the larger network — meeting new FDA guidelines that recommend that if a device has invalid software, it can detect and report it.
Rule 3: Protect critical data. Patient data, key operating parameters, and even software need to be protected not just in transit over the network, but also within the device. This is accomplished by a security design that incorporates separation and encryption to ensure that only authenticated software and users have access to stored data.
Protecting data in transit requires that data can only be viewed by the proper endpoint. Note that standard wireless encryption does not provide secure communication; it protects the data link only, but not the data. Any other system that can access the wireless network is also able to view the packets in decrypted form. Data protection is accomplished through network security protocols, such as TLS, which enables secure client/server communication through mutually authenticated and uniquely encrypted sessions.
Rule 4: Secure keys reliably. Keys used for encryption and authentication must be protected, because if these keys are compromised, an attacker may uncover sensitive data or emulate a valid endpoint. For this reason, keys are isolated from untrusted software. Keys stored in nonvolatile memory should always be encrypted, and only decrypted following secure boot verification. Since protection of patient data is paramount, especially for the EU, use of high-assurance kernels and security modules also provide layered separation for fail-safe design.
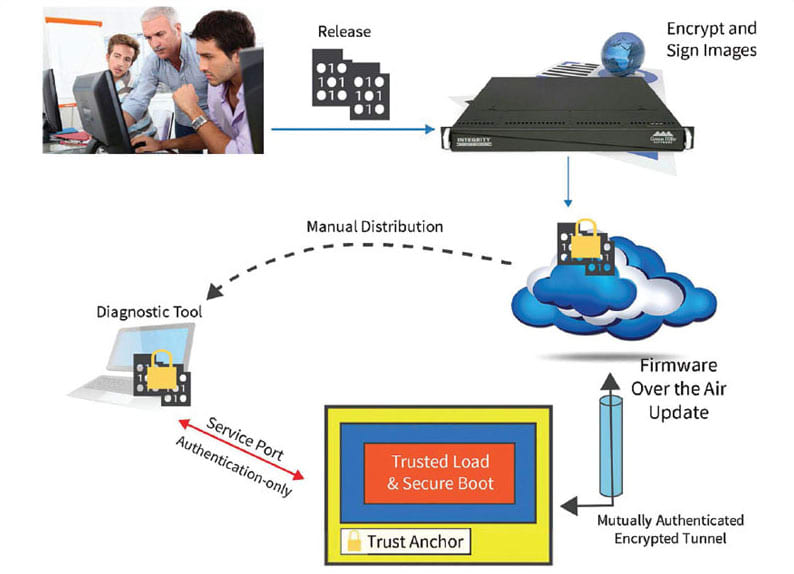
Keys need to be protected in manufacturing and throughout the product life cycle by an end-to-end security infrastructure. If a key is readable at any time, all of the devices using it are vulnerable. An enterprise security infrastructure protects keys and digital trust assets across distributed supply chains, but can also provide additional economic benefits beyond software update such as real-time device monitoring, counterfeit device protection, and license files to control availability of optional features (see Figure 3).
Rule 5: Operate reliably. As all medical designers know, among the biggest threats to a system are unknown design errors and defects that can occur during the development of complex devices. To address these potential threats, it is a good idea to implement principles of high-assurance software engineering (PHASE). These principles include the following:
- Minimal implementation — Code should be written to perform only those functions required to avoid “spaghetti code” that is not testable or maintainable.
- Component architecture — Large software systems should be built up from components that are small enough to be easily understood and maintained; safety and security critical services should be separated from noncritical ones.
- Least privilege — Each component should be given access to only the resources (e.g., memory, communications channels, I/O devices) that it absolutely needs.
- Secure development process — High-assurance systems, like medical devices, require a high-assurance development process; additional controls beyond those already in use, such as design tools security and secure coding standards, may be needed for ensuring a secure design.
- Independent expert validation — Evaluation by an established third party provides confirmation of security claims. It is also often required for certification. As with functional safety, components that have already been certified for cybersecurity are preferred as reliable building blocks in a new design.
These principles are used in the development of Green Hills Software’s INTEGRITY real-time operating system. When applied to application development, they minimize the likelihood and impact of a software error or a new cybersecurity attack.
Creating an End-to-End Security Solution
Building a secure medical system that meets the new regulatory environment requires an end-to-end security design that addresses the security of data and reliability within the networked device throughout the product life cycle. This requires a device security architecture, which safeguards operation by ensuring that keys, certificates, and sensitive data are protected throughout operation and manufacturing supply chain by an enterprise security infrastructure. The optimum selection of both device and enterprise security solutions depends on device operating and manufacturing environments, as well as business tradeoffs, so it is advisable to consult experts in the field.
This article was written by Mary Sue Haydt, Field Applications Engineer for Green Hills Software, Santa Barbara, CA. For more information, Click Here .



