Obtaining detailed images of cancer cells buried deep within normal tissues has slowed the usefulness of existing imaging technology. An approach biomedical engineers are pursuing to overcome this limitation involves building nanoprobes that are designed to travel throughout the body, accumulate in cancer cells, and send a signal that illuminates the tumor.
NIBIB-supported researchers at Johns Hopkins University have developed a smart nanoprobe designed to infiltrate prostate tumors and send back a signal using an optical imaging technique known as Raman spectroscopy. The new probe has the potential to determine tumor aggressiveness and could also enable sequential monitoring of tumors during therapy to quickly determine if a treatment strategy is working.
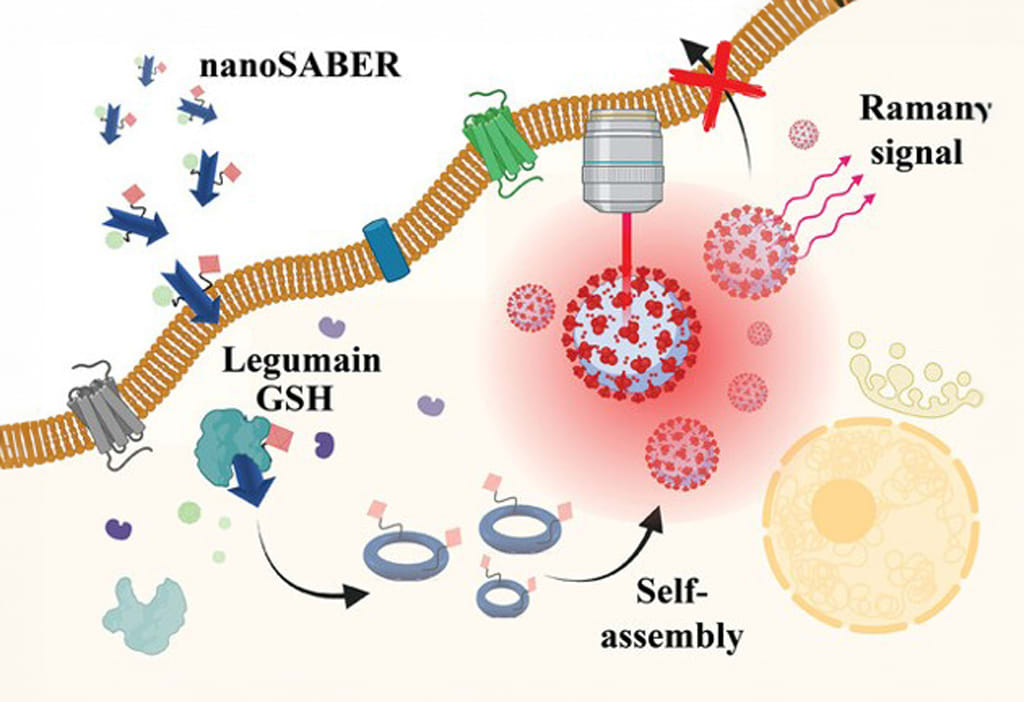
The engineering team constructed a nanoprobe that is sensitive to its local microenvironment: it is activated only after it encounters legumain, a tumor-associated enzyme that is produced by aggressive prostate cancer cells. Once it encounters the nanoprobe, legumain splits it into pieces that can self-assemble to create an optically active nanoparticle. These nanoparticles emit specific wavelengths of light that can be detected with Raman spectroscopy to visualize the tumor.
The researchers used two prostate cancer cell lines exhibiting varying levels of legumain expression — one high and one low — alongside a non-cancerous prostate epithelial cell line, which produces a negligible amount of the enzyme.
NanoSABER was tested in laboratory cell cultures and in an experimental mouse model. In both settings, the prostate cells expressing legumain activated the nanoprobe and emitted a signal with an intensity that corresponded to the amount of legumain produced by the cancer cells. The non-legumain cell type did not activate the nanoprobe, demonstrating that the nanoSABER system performed as designed, emitting a signal that correctly indicated the presence and amount of the cancer-associated enzyme.
The team believes they have engineered a molecular system with the potential to not only identify tumors using optical imaging but to also rapidly assess tumor aggressiveness — potentially without the need for painful biopsies that are the current standard of care. In addition, as profiles of enzyme secretion by different types of cancers are discovered, additional nanoSABER probes can be synthesized that will allow a level of precise diagnosis of tumor types and characteristics that is not currently possible, including sequential imaging of tumors to determine whether therapies are working in real time.



