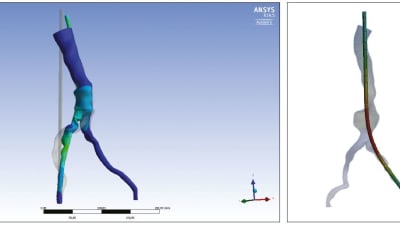
Features: Medical
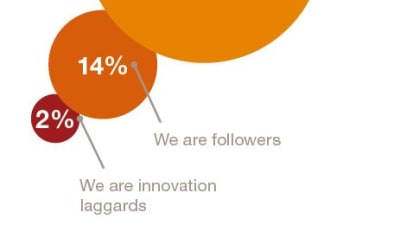
With the US innovation ecosystem widely perceived to be under severe stress, both government and business leaders are actively reviewing their options for meeting emerging market needs.
Features: Electronics & Computers
Today’s medical device and equipment designs are highly influenced by continuous technological advances that affect their size, power consumption, and communication capabilities....
Features: Medical
Grand Prize Winner
ChemoPatch
Alydaar Rangwala, Nikhil Mehandru,Aaron Perez, and Brandon SimTheratech, Loudonville, NY
Existing methods for administering chemotherapy...
Features: Materials
Polycarbonate (PC) is one of the engineering thermoplastic materials most commonly used and most widely tested in the medical device industry today. Its inherent strength,...
Features: Manufacturing & Prototyping
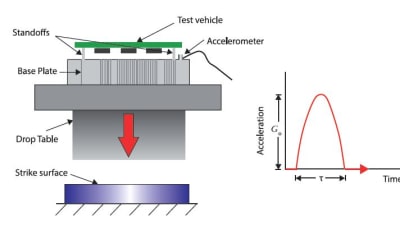
Stringent product quality and process capability requirements confront producers of precision medical tubing used in medical device applications. This includes final tubing physical...
Features: Materials
Additive manufacturing, also known as 3D printing, grew from startup roots in the mid- 1980s to a $2.2 billion business by 2012, according to industry consultants Wohlers Associates,...
Features: Manufacturing & Prototyping
If molten plastic behaved like a simple fluid, there would be little need to worry about balanced filling during molding. The melt would fill the cavities like water, and the way the mold filled...
Features: Materials
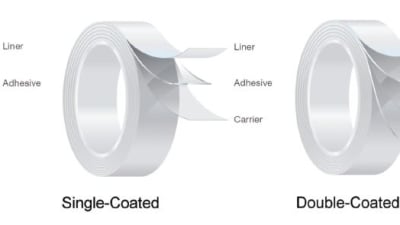
Medical device designers frequently face the need to adhere products to the skin, whether it is for a few minutes or in perpetuity. The huge array of pressure-sensitive materials...
Features: Medical
Over the years, medtech companies have become quite adept at working with clinicians to identify unmet clinical needs, and developing products to address those needs. In both start-ups and established...
Features: Manufacturing & Prototyping
A growing array of electronic devices are available to healthcare providers, patients, and their families, including glucose meters, blood pressure...
Features: Electronics & Computers
History
Although the deadline for RoHS compliance for medical device manufacturers is nearly a year away (July 22, 2014), there is more than ten years of testing and in-service data that can be...
Features: Manufacturing & Prototyping
Around the world, people are living longer, more active lives thanks to continuing advancements in medical technology. This evolution in technology stems from the development of...
Features: Tubing & Extrusion
Medical device manufacturers are being challenged by strong market demand for tubing that delivers increased functionality, lower profiles, and lower costs—pushing the limits of material...
Features: Medical
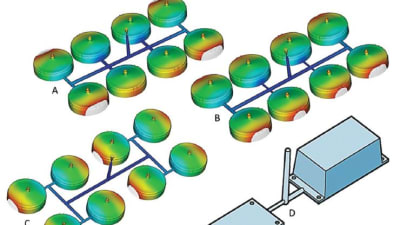
Medical imaging technology is improving at a remarkable speed, but most imaging technicians and physicians still use a mouse and keyboard to manipulate in two dimensions the complex ultrasound, computed...
Features: Medical
Recent demands from hospitals, practitioners, and even patients themselves calling for more functions and increased portability, has created a market for upgraded surgical,...
Features: Software
A newborn is diagnosed with a heart condition called hypoplastic left heart syndrome in which the left ventricle of the heart is severely underdeveloped, and requires immediate surgery. While there...
Features: Medical
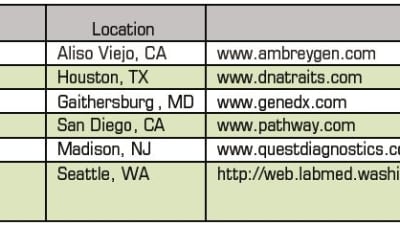
In June, academic researchers and clinical laboratories seeking to make a mark in the emerging fields of molecular diagnostics and personalized medicine were buoyed by the long-awaited ruling of the U.S. Supreme...
Features: Medical
Insulating and jacketing material options for wire and cable are innumerable, even if the field is narrowed to those with some qualification for use in medical electronics....
Features: Medical
What is DFx? DFx is an acronym used often in reference to concepts or guidelines involving design for manufacturability, test, and production readiness. In the world of innovative and complex medical devices, DFx can...
Features: Motion Control
As an increasing number of patients enter the operating room, more and more orthopedic surgeons are becoming orthopedic patients themselves. According to a survey entitled “Occupational Hazards...
Features: Medical
Managing the environmental and regulatory performance of products is an increasingly complicated challenge for medical device manufacturers who face a myriad of requirements from regulators,...
Features: Medical
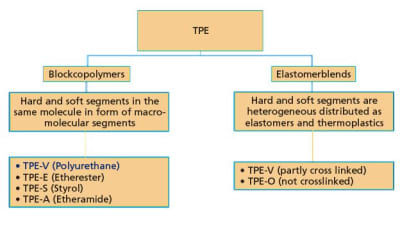
Thermoplastic polyurethane (TPU) is well known and specified in the medical industry for advanced medical and healthcare products, due to its high performance characteristics. Because of...
Features: Medical
Hospital-acquired infections (HAIs) are a major area of concern for providers, payers, and patients alike. These infections play a significant role in the estimated...
Features: Medical
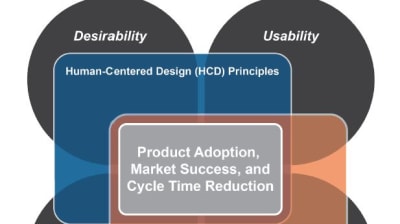
Developing a medical device requires sensitivity to the delicate balance between usefulness, usability, desirability, and manufacturability. Every medical device must be useful (meet a need) and...
Features: Information Technology
Over the past decade, the power of physician preference has steadily diminished in the face of hospital and physician consolidation, continuing pricing pressures, and structural changes in payment systems...
Features: Medical
Introduction
A well-known legend has it that one of the greatest scientists and inventors of antiquity, Archimedes of Syracuse, stepped into a bath only to eject and propel...
Features: Sensors/Data Acquisition
Electrocardiogram (ECG or EKG) measurements typically involve time-consuming skin preparation, lead application, conductive gels, and even shaving of body hair. More recently, dry contact sensors...
Features: Electronics & Computers
Today’s medical device OEMs live in an age of abundance: both abundant options and abundant regulatory requirements. With so many options and so many restrictions, it can be very...
Features: Regulations/Standards
Now that the Centers for Medicare and Medicaid Services (CMS) has published its long-awaited final rule implementing the Physician Payments Sunshine Act (Federal Register, February 8, 2013), medical device manufacturers can move forward to carry out a familiar task: complying with regulations.

 Podcasts: Robotics, Automation & Control
Podcasts: Robotics, Automation & Control Podcasts: Design
Podcasts: Design Podcasts: Medical
Podcasts: Medical Podcasts: Design
Podcasts: Design Podcasts: Medical
Podcasts: Medical On-Demand Webinars: Communications
On-Demand Webinars: Communications