Imagine patients with chronic conditions being monitored without having to be in a doctor’s office, or patients with more serious issues being monitored and treated outside the intensive care unit in a much less costly hospital room — or even at home. That’s the promise of wearable medical devices, a market that has been around for decades, but is being re-energized by new technology, including wireless communication.

The latest generation of medical wearables, when fully developed, will not only offer more patient convenience, but also the capability to drastically reduce medical costs. By eliminating the need for expensive in-hospital monitoring or potentially eliminating the need for costly medical procedures by providing early detection and diagnosis, medical wearables may dramatically improve the delivery of medical services. Also, healthcare reforms are driving compliance to prescribed therapies to ensure a positive outcome, and miniature wireless devices can fill this gap.
Medical wearables also have the potential to provide better-quality data and improved monitoring compared to large, expensive machines located in hospitals and research units. For example, today, sleep apnea patients or research subjects must come into a clinic and be wired for sleep studies. However, people generally have trouble sleeping in rooms other than their own. A wearable medical device that could monitor sleep apnea metrics in a much less intrusive way could obtain much more reliable, higher-quality, and longer-term data to diagnose patients and study the effects of the disorder.
Also, consider a condition like epilepsy where known physiological changes that occur in people over time simply can’t be measured 24/7 with today’s equipment. With a wearable device, a patient could be monitored for an extended period of time, providing a much better understanding of how they interact with their environment and the medicines they take. That information could potentially be used to develop a predictive capability that warns a patient when they’re about to have a seizure, or if they are at a higher probability of having a seizure.
Types of Devices
There are four primary types of wearable medical devices, ranging from the simplest to the most complex: passive monitors, surveillance devices, diagnostic devices, and therapeutic devices. Of the latter three, surveillance is the simplest. Moving along the continuum from surveillance to therapeutics, barriers to commercialization increase. Surveillance devices simply report, while diagnostic devices make decisions, and therapeutic devices make decisions and treat the patient.
Diagnostic devices carry a higher level of legal risk than do surveillance devices because they find problems and medical decisions are being based on the data they produce. Therapeutic devices take that risk to an even higher level because they treat the condition. Most therapeutic wearable devices are still in the research phase, and would have to be approved as medical devices by the FDA, making the development cycle much longer — particularly if a company is developing a drug/device combination. Monitoring devices make sure that diagnostic and therapeutic decisions and procedures have been done correctly.
Development and Design
Two key factors in wearable medical device design are ease of use and patient comfort. They are typically determined by how the electronics are configured and what type of materials are used to fabricate the electronics. Use of rigid materials must be minimized so the device can conform to the body and survive bending during normal human motion.
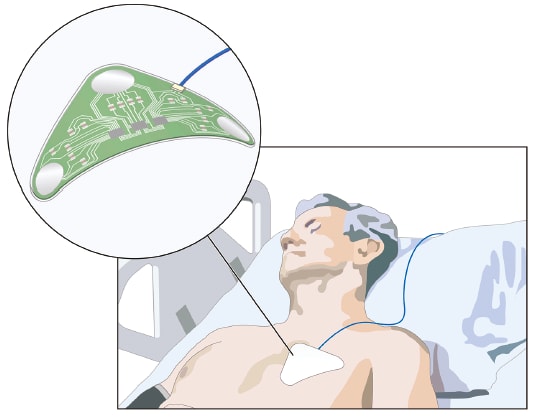
As a result, designers and component suppliers must first understand the use scenario. For example, where would the wearable device be applied — wrist, chest, elbow, lower back, or elsewhere? How will it be used — once a day and then replaced, or for a week or more before being replaced? Understanding use scenarios includes asking questions such as:
- How does the device need to function electronically?
- Will the devices include wireless interfaces to support telehealth and mHealth applications?
- At what intervals does it need to collect data for the different metrics being monitored?
- How is that data stored? Does the device access a local storage site, or does it communicate with the cloud?
Because skin contact is involved in most wearable applications, special adhesives or other fastening means must be included in the design. The adhesives must not cause any skin irritation and should account for varying patient sensitivities. Fortunately, a wide range of biocompatible medical adhesives can be used, with the choice of adhesive dependent on a number of factors, including the length of time the device is going to be worn; for example, for 15 minutes or seven days. Another consideration is if the wearer will be exposed to water or if they will shower with the device on.
Types of Electronic Circuits
The choice of electronics for medical wearables typically includes traditional PCBs, copper flex circuits, printed electronics, antennas, fine wires, or combinations thereof. PCBs, being rigid, are typically appropriate for applications such as smart watches and wellness-type devices that tolerate the form factor and its rigidity.
Flexibility is a key advantage of etched copper flex circuits because it enables improvement in form factor relative to rigid PCBs. Complicated components such as microcontrollers with high I/O counts and other fine-pitch devices can be attached to copper flex substrates. While these packaged devices limit the flexibility and conformability of the overall circuit, they can meet many performance requirements.
Copper flex circuits are fabricated in a subtractive process by taking sheets of copper laminates, masking the desired conductive paths, and then chemically removing all unwanted copper, leaving the desired circuit patterns. This is followed by component attachment, as discussed above. While copper flex circuits remain a key part of many medical devices, in certain applications, they may have issues with biocompatibility as well as failure due to repeated bending cycles.
Additive manufacturing techniques are used to create the base circuit in printed electronics. Typically, silver, silver chloride, carbon, and dielectric materials are printed on a variety of substrates that can be chosen for biocompatibility. The materials and the manufacturing process contribute to making base circuits that are extremely flexible and able to withstand many bend cycles. In addition, certain substrates and printed materials can be stretchable, offering additional robustness for applications where patient motion is involved. The substrates and manufacturing approach for the base circuits can often provide cost benefits compared to copper flex circuits.
As with copper flex circuits, components can be attached to the printed electronic base circuits, enabling high-performance devices. As a result, many wearable medical device designers are evaluating printed electronic solutions to leverage these advantages.
Choosing the right circuits for a medical device is not a zero sum game. There are some wearable applications where a designer could combine all three types of circuits, or use copper flex circuits and printed electronics in the same package to provide functionality and still meet the device’s cost target.
Converting and Packaging
There are many different converting techniques for wearable medical devices, and the choice depends on how the final product should look and feel. Medical converting can be performed in sheet-based or roll-based forms. The latter is generally more applicable to higher-volume products. Also, roll-to-roll medical converting substantially reduces the labor component of converting and packaging, and multiple rolls of materials can be joined and laminated together on the machine.
The converting process can include die-cutting, laser cutting, slitting, island placement, and lamination — all in a continuous operation in the case of roll base converting. Often, multiple units of the same product are created on each sheet in the case of sheet-based converting, or on repeat length in the case of roll-based converting. Those products are then singulated into individual products, packaged, stored, and shipped.
Roll-to-roll converting can also be combined with more costly sheet converting. After beginning medical converting on a roll-to-roll process, the rolls can be sliced into sheets, with the final converting steps performed on a sheet-to-sheet basis. Sheet-based converting is non-continuous and throughput is typically lower than in roll-to-roll converting. However, some sheet-based converting processes can be more accurate than roll-to-roll. Choosing the right process for a particular wearable depends on required throughput, accuracy, device cost, and other factors. For example, if a manufacturer is performing roll-to-roll medical converting and device volume is not high enough, scrap and setup will drive unit costs higher. In that case, a sheet manufacturing process might be more appropriate.
Software Component
Software is another key wearable medical device component. The device manufacturer typically has the information needed to develop the software, including intimate knowledge of the patients, the type of physiological metrics being monitored, how changes in metrics affect the patient, and how that data can be used to inform caregivers.

Because wearable medical device analytics is a relatively new field, software developers are creating systems that can translate patient knowledge into new algorithms and data analysis for highly specific applications. Also, antennae and mobile technologies can be part of complete wearable medical solutions — a product group that includes smartphone-controlled devices. Close collaboration among device manufacturers, software developers, and electronics providers is essential to make sure software meets the needs of the user.
For example, physiology analytics can be used to analyze the relationship among between physiological signals such as blood pressure, heart rate, respiration rate, and others, providing continuous monitoring and alerts when needed.
Using Manufacturing Partners
When all the pieces are in place, there are several options for final wearable device assembly. If the developer controls all aspects of the design, a contract manufacturer can build the device based on design specs. On the other hand, most wearable medical devices require a collaborative design process that includes manufacturing partners.
In those cases, it is essential for device manufacturers to work with partners on all aspects of device commercialization, from design through manufacturing. Most wearable device marketers do not manufacture the electronics that power these devices, so partners typically play an outsized role in developing and manufacturing the devices. The partners help customers design devices, move them through prototypes stages, select the most appropriate technology, and support them through regulatory approval by the FDA or other regulatory bodies.
In the case of wearable medical devices, which are relatively new products, a great deal of expertise resides outside of the device manufacturer. There is a long history of devices being manufactured for medical companies by manufacturing partners. What these partners can bring to the table is a deep understanding of how devices need to function and interact with the human body, as well as the required manufacturing techniques and how to optimize and combine them appropriately to provide the most efficient manufacturing solution. This synergistic relationship can provide a critical comparative advantage for device manufacturers that want to bring the best possible solution to the market.
This article was written by John Heitzinger, Industry Marketing Manager at Molex, Lisle, IL. For more information, Click Here .
Prototype Wearable Device Measures Lactate in Sweat
People in stressful, physical situations sweat, and that sweat can speak volumes about what their bodies and minds are going through. Molex, Northeastern University, Brewer Science, and American Semiconductor are developing a wearable medical device that can analyze sweat to provide physiological clues about the well-being of the people being monitored. Funded by the U.S. Air Force, the wearable patch will measure the presence of biomarkers such as lactate in human sweat. The noninvasive patch would collect and analyze sweat, then provide that data via a user interface to either a caregiver or the device wearer, depending on the application. When the device is fully developed, the patch would likely communicate to a cellphone or other mobile device also worn by the subject that could then communicate to the cloud wirelessly, though that portion of the project is not currently under development. The partners are currently working on a developmental prototype.



