A healthcare-acquired infection (HAI) is commonly defined as any infection that is contracted while receiving any form of healthcare. HAIs are a seemingly unavoidable consequence of the modern healthcare system, which is designed to house and treat the most at-risk populations. Especially during times of crisis such as the current COVID-19 pandemic, surging hospital admissions raise concern that HAIs may also be on the rise.
According to the CDC, nearly 1.7 million people acquire HAIs every year, and 1 in 17 of them die from the infection.1 The World Health Organization has confirmed this to be a global issue, citing evidence that hundreds of millions of patients are affected. 2 Collectively, these organizations have offered their best practices for addressing the root causes of the issue, suggesting that the increased use of disposable products can make a significant impact in reducing the number of infections. This push has created a boom in the medical disposables industry, resulting in a 10 percent CAGR.
Medical disposables are single-use products that can include everything from gloves to face masks, needles, and blood bags. These products can replace equipment that typically undergoes a time-consuming and costly sterilization procedure, despite still resulting in frequent HAIs. Despite conventional knowledge suggesting that reusable products would be more expensive over time, the cost of the sterilization procedure coupled with the additional costs associated with extended hospital visits offsets the bulk of the expense.

While the move toward disposable medical products has helped to partially curb the spread of HAIs, it has exacerbated another issue — the mounting environmental concerns over healthcare waste. Hospitals are one of the largest producers of waste, much of which needs to go through complex processing to neutralize toxic or infectious byproducts. WHO estimates that in high-income countries, approximately 0.5 kg of waste is produced per hospital bed per day.3 As a result of national and international guidelines, many healthcare facilities are putting heavy emphasis on reducing their carbon footprints, highlighting the importance of reevaluating their waste management systems and recycling programs. Waste that can be categorized as non-toxic represents the largest opportunity for a potential solution.
Faced with these two obstacles — a need for single-use disposables and a simultaneous waste management crisis — the healthcare industry is being forced to innovate. Using new materials and processes can help reduce the environmental impact of hospitals while still driving down the rate of HAIs. The use of biodegradable materials is emerging as a clear path forward, providing a viable alternative for traditional materials like plastics and textiles. Biodegradable materials, and specifically plastics, are typically produced using more environmentally friendly means compared with traditional plastics, ensuring that the entire product life cycle has a significantly smaller carbon footprint (see Figure 1). Companies around the globe are innovating to develop novel materials to address the issue, engineering materials from corn, algae, mushrooms, and a whole host of other natural sources. To expedite this shift, material testing is essential for validating the performance of these new materials and ensuring that their specifications are comparable to existing materials.
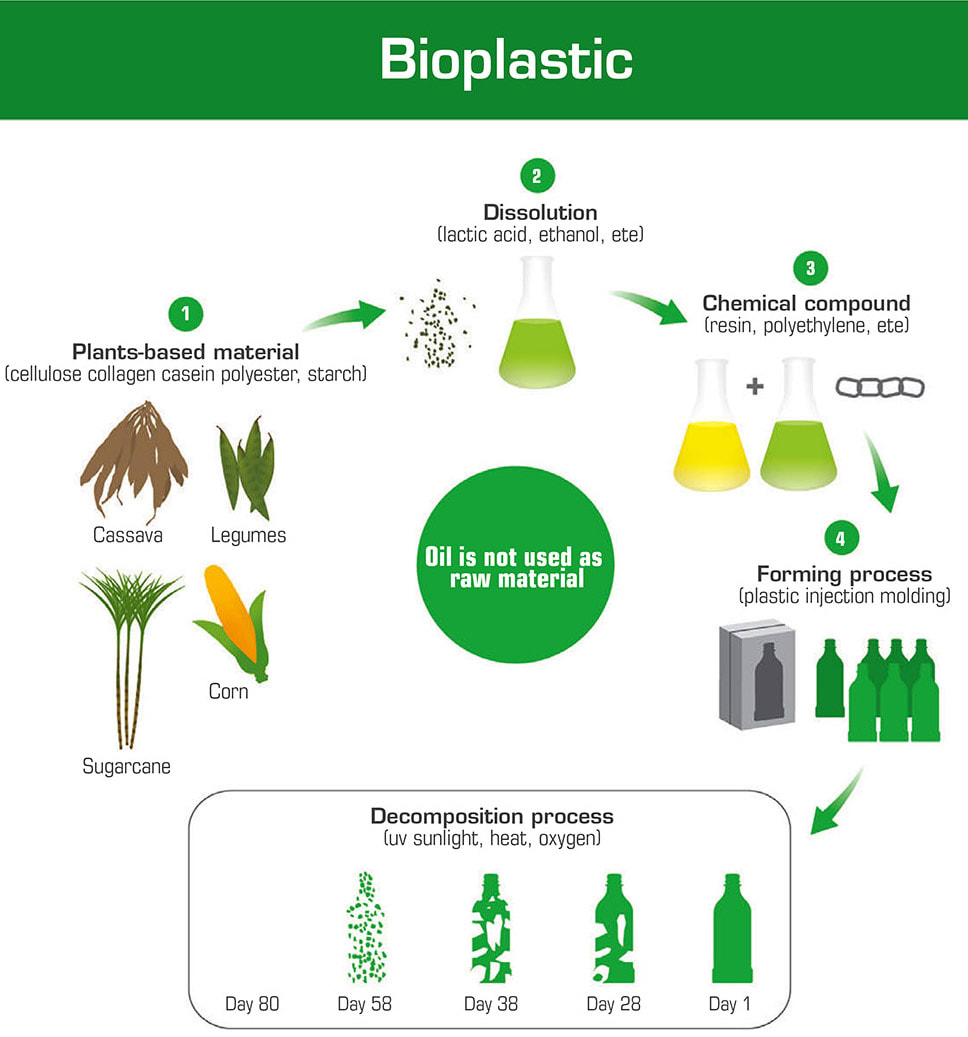
The development of bioplastics has become a burgeoning industry with a strong market need, especially in the healthcare industry. Within the hospital setting, plastics can be used in consumables like syringes and tubing, medical-grade gloves, and even trays and cups used by patients. Each product represents an opportunity to reduce the amount of non-degradable waste produced by the hospital.
Two main types of bioplastics have dominated this infant market — PLA [polylactic acid] and PHA [polyhydroxyalkanoate]. PLA is produced using the sugars found in corn starch, cassava, or sugarcane. Depending on the specific manufacturing process, the end product can be made to mimic traditional plastics like polyethylene, polystyrene, and polypropylene. PLA is especially promising due to its biocompatibility — making it a natural candidate for applications in medical implants, orthopedic devices, and drug-delivery systems. PLA has even played an important role in the global pandemic, being utilized as 3D printable polymer for ventilator components and in the form a filament to manufacture personal protective equipment (PPE). 4
PHA is a biopolymer that is actually created by microorganisms that convert organic material into a polymer byproduct. In its harvested state it can be utilized for many medical consumables, most notably for surgical sutures and meshes. Taking a broader view of hospital waste, PHAs are ideal candidates to be used for single-use food-grade packaging. These are just two of the most common bioplastics being produced today, but research is ongoing to identify alternative materials that balance performance and degradability. Hospital-wide initiatives and leadership-backed support will be required to supplant the use of traditional plastics with bioplastics in a range of end use applications.
It is worth taking some time to briefly define the terminology related to degradability. One might intuitively assume that all biopolymers are able to be composted, but this is not necessarily the case. Most experts place all plastics and bioplastics into three categories of degradation: degradable, biodegradable, and compostable. Even traditional plastics can be defined as degradable, which simply means that over some amount of time and the appropriate environmental conditions, they will disintegrate into microscopic fragments. This is the source of the microplastics that have become a serious issue for most bodies of water.
Most bioplastics fall into the category of biodegradable, but there are still some gray areas, as the term does not have an agreed upon set of requirements. Typically, biodegradable means that microorganisms like bacteria and certain fungi can convert the material into water, carbon dioxide, methane, biomass, and inorganic compounds. This process can usually occur within a few months, but those that take longer are given the designation of durable bioplastics. The term compostable refers to bioplastics that are converted completely into nutrient-rich biomass with no toxic byproducts. As an example, PLA can be composted and will produce 70 percent fewer greenhouse gases as it degrades than petroleum-based plastics.5 Globally, standards are being developed to better define these terms. Both ASTM D6400 and ISO 17088 include definitions for compostability, for example.
Validation and Testing
The biomedical industry is easily one of the most highly regulated in the world and, as such, significant validation and testing is required to introduce a new product or even utilize a new material for an existing product. Standards organizations like ASTM and ISO typically lag behind innovation because of the stringent evaluations and the consensus required by a consortium of industry experts. This process is necessary to create universal standards to meet the needs of manufacturers and their labs. Currently, standards are in development to address a range of biopolymer properties across many medical and consumer product applications.
In the meantime, many typical plastics standards like ASTM D638 or ISO 527 for tensile properties have updated their verbiage to broaden the scope to include bioplastics and 3D printed plastics. The testing requirements for these standards are well known, and by using them for evaluation, researchers get a clear correlation in material properties between petroleum and biologically based plastics. To fully characterize a material, depending on the end use, a researcher may need to perform tensile, compression, flexure, torsion, and a myriad of other test types. R&D facilities need the tools at their disposal to comprehensively analyze these materials to eventually replace traditional plastics and clear the regulatory hurdles that FDA and other international organizations will create. Universal testing systems can offer a flexible solution to meet the challenges of this new frontier.
Can the use of bioplastics completely eliminate these two crises facing the healthcare industry today? On its own, no, but the gradual replacement of traditional plastics coupled with more robust waste management programs and the introduction of forward-thinking environmental policy in the healthcare industry can significantly reduce the negative impact made by plastic waste in the last 50 years. The data has shown the sizeable effect disposable products can have on reducing HAIs. The development of biodegradable and compostable plastics addresses the other side of the issue.
As these materials become more advanced and are adapted to new applications, material testing will serve as a proving ground, both evaluating physical properties and even discovering potential uses previously unknown. Scientists will need to be equipped with the tools necessary to assess and validate these products, requiring experienced equipment suppliers ready to face these technical issues head on. Looking at the larger picture, the biomedical industry represents only a fraction of the potential for these polymers, but early adoption will allow it to be at the forefront of a paradigm shift in not only the plastics industry, but the entire global emphasis on sustainability.
References
- Haque, M., Sartelli, M., McKimm, J., & Abu Bakar, M. (2018). Health Care-Associated Infections – An Overview. Infection and Drug Resistance, 11, 2321–2333.
- (2011). Report on the Burden of Endemic Healthcare-associated Infections Worldwide. Geneva: World Health Organization.
- World Health Organization. (2018). Healthcare Waste Fact Sheet. Retrieved from here.
- DeStefano, V., Khan, S., & Tabada, A. (2020). Applications of PLA in Modern Medicine. Engineered Regeneration, 76–87.
- UrthPact. (2018, March 29). The Basics of BioPlastics. Retrieved from here.
This article was written by Landon Goldfarb, Senior Applications Engineer, Instron, Norwood, MA. For more information, visit here .




