The medical industry is behind the times when it comes to user-centered design and user experience. Healthcare professionals are demanding a consumer product level experience. “Make it like my iPhone” is a phrase we have all heard. UX (user experience) positions and UX boot camps are starting to emerge in large medical companies to solve this problem but plugging in a prescribed process won’t cut it. When it comes to medical device design, the stakes are higher, the environments more extreme, and the technology more complex, not to mention the reign of regulatory.
UEGroup, for example, specializes in making medical devices intuitive and safe, taking groundbreaking tech and making it approachable for users.
This includes beefing up the user centered process, injecting more evidence-based iteration. Here are five medical device specific strategies UEGroup recommends to make stronger products with stronger FDA submissions.
1. Test the Wrong Thing to Learn the Right Thing
This may sound ridiculous. Why waste time testing something that won’t work in the long run? The answer is simple: we learn from our failures, and this first failure is a critical one. When you save testing for something close to the real product, you are getting users opinions on something that has already been formed. It has been built on assumptions and the team’s interpretation of the trade-offs without the voice of the user.

Instead of starting with assumptions, we recommend understanding the users’ perspective of these trade-offs prior to making anything real. When trying to understand the correct balance between trade-offs, we like to show the extremes. We know that an extreme example is not going to be the final design, but we want our users to tell us that and, most importantly, we want them to tell us why. Oftentimes these extreme solutions lead to much richer conversations and understanding of their mental models. With this method, we can rule things out based on evidence and pick a path forward combining successful elements in line with user priorities and thought process. This removes the assumption from the design foundation and replaces it with evidence making a stronger product and process.
2. Prioritize Simplicity over Complete Freedom and Flexibility
Complete freedom is required in more exploratory or browsing based experiences like streaming services, e-commerce, and even file management systems. Things change once a product shifts into a task-based experience when efficiency is the driving factor. For medical devices, you add the requirements of error prevention and clarity to ensure user confidence and quick reaction times needed in critical situations. In these cases, it is important to optimize the interface to a single task at a time, highlighting a step of a precise workflow.

Status, primary controls, and navigation must be obvious and easily accessible, but all other screen clutter is just a distraction a user has to sort through. That being said, stripping back too far can be confusing for users. Labels and iconography that work together is most preferred by medical staff. We encourage a single task in a larger workflow, but multitasking and flexibility to switch course are required in the OR. We recommend supporting this in the navigation structure, so it does not take away from the simplicity of a single page.
3. Test with the Nonusers for Basic Usability
It can be expensive and time consuming to test with medical professionals, especially if you’re doing full cadaver labs. These labs are very informative and important part of the process, but they should be saved for procedure and specialty driven questions. A major lab and full surgeon set are not necessary for general usability questions, and often-times, surgeons get annoyed if you ask them to do basic usability tasks.
Save your surgeons and money. General questions like touch interactions, discoverability, color usage, etc. can all be done with average people. These elements can also be more subjective, so having a larger participant pool becomes more important for reliable data. We have done tests using both nurses and nonmedical participants and the results were consistent. Running tests this way is cheaper, faster, and produces more data.
4. Don’t Be Afraid to Challenge Regulatory
Unlike consumer products, medical designers need to be complaint with regulatory standards. In these guidelines, there are specific use instructions for color, font sizes, and messaging, but following these to a T does not guarantee a strong usable product. One area that has a bit of conflict is messaging. The guidelines require a specific pop-up message for information, warning, and error messages. From a regulatory perspective, this is the safest form of message because a user has to read it and take an action addressing the message.
However, if every message in your system appeared as a pop-up, users would be throwing things out of frustration. When the guidelines don’t make sense for your product scenario, don’t use them. Instead of strictly using pop-ups, introduce more messaging types seen in consumer products. Use a banner messaging model for informative messages that should not interrupt the user.
If a banner doesn’t draw enough attention but a pop-up still isn’t justified, try something more dynamic. Capitalize on motion and consider the user’s area of visual focus. Banner and dynamic messaging models are not in the regulatory guidelines, but that is ok. The guidelines are a starting point and you can break from them as long as you can prove effectiveness in user testing and the change cannot lead to patient harm.
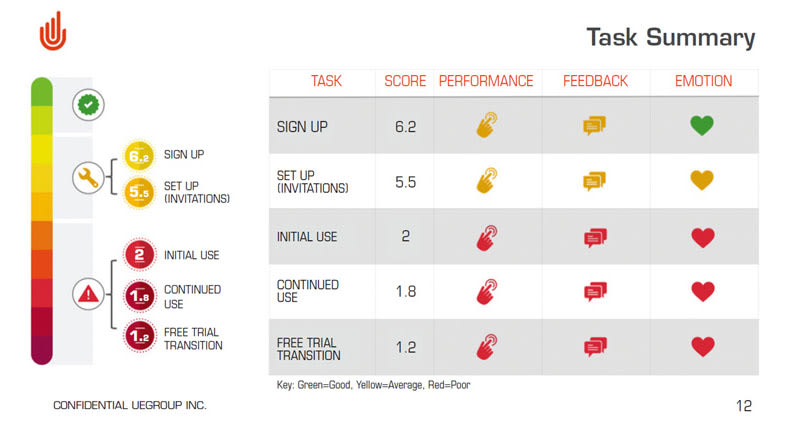
5. Use Success as a Metric
All projects have goals. At UEGroup we like to incorporate these goals into our testing, so we can track our progress through iterations. We start by identifying the facets of success to the product and then convert that into quantitative ratings.
Let’s say our goal was to create intuitive software that a user will feel confident interacting with. Then we would use the Pass/Struggle/Fail metric and a scale question “How did you feel completing the task?” At every iteration, we would collect these metrics and see whether the changes have pushed us closer or further from our goal. By the time a final design has been reached, the team does not need to convince the larger organization that the product reached its goals. There is proof through user feedback and benchmarking that the design is correct.
This article was written by Ashley Nicodemus, designer for the UE Group, San Jose, CA. For more information, visit here .