On Sept. 21, 1960, Dr. Albert Starr successfully implanted the world’s first “ball-in-cage” prosthetic heart valve into a patient’s mitral valve, a mechanical device that he co-invented with M. Lowell Edwards, a prominent engineer in the biomedical field. The artificial valve was a great success in the medical device industry that paved the way for numerous developments in cardiology. More than 50 years later, millions of patients’ lives have been saved or improved by the technological advancements based on the work of pioneers like Dr. Starr, Edwards, and others. Every year, more than 250,000 patients worldwide receive prosthetic heart valves and that number continues to grow.1
Heart valves play an important role in directing blood flow and preventing back flow in the heart. Today, there are two types of artificial valves available: mechanical heart valves (MHV), such as the Starr-Edwards device, and biological valves made from tissue material, also known as bioprosthetic heart valves (BHV). There are advantages and disadvantages to both types. Mechanical valves have superior durability and can last a lifetime, but they can cause blood clots with life-threatening consequences, requiring patients to take blood thinners for the rest of their lives.2 Bioprosthetic heart valves have excellent hemodynamics that reduce the risk of blood clots. However, they wear out and usually require replacement within 10 to 15 years in older patients and much sooner in younger patients.
Design and development of BHV has been a major focus of research in the field of cardiovascular engineering. BHV is a better replacement because it causes fewer side effects and can closely mimic natural flows, compared to MHV. While patients who undergo heart valve replacement experience a better quality of life, complications can occur due to device failure, damage to the valve caused by infection, poor valve design, material deterioration, or host tissue overgrowth.
Bioprosthetic heart valves tend to calcify around the tip, potentially as a result of excessive stress. This calcification can lead to fatigue, tearing, and ultimately valve failure if the valve is not replaced. By reducing the stress and subsequent calcification, researchers can improve the longevity of the BHV, making it a better replacement option for patients.
Visualizing a Better Bioprosthetic Heart Valve
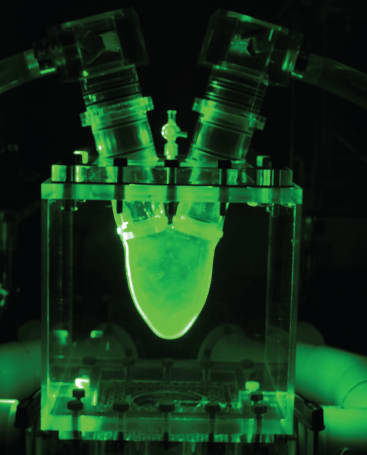
Ahmad Falahatpisheh, a Ph.D. candidate in the lab of Prof. Kheradvar (KLAB) at the University of California, Irvine, is pioneering research to improve bioprosthetic heart valves and patients’ quality of life. Using scientific visualization software to view and analyze his Digital Particle Image Velocimetry (DPIV) data, Falahatpisheh is helping to develop the first bileaflet bioprosthetic mitral valve with a dynamic saddle annulus designed to mimic the natural mitral valve.
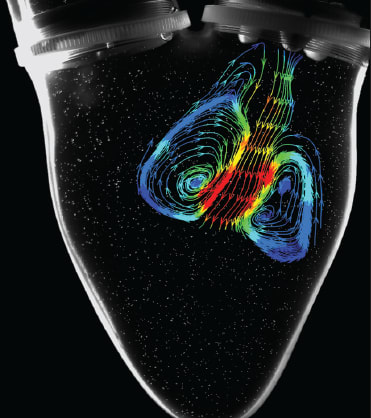
Flow features such as vortices developed in this valve are very similar to its natural counterpart. In addition, stress at the tip of the leaflets, which can cause leaflet damage, is much less than the valves with rigid annulus. This is mainly due to the dynamic annulus made of Nitinol, which adapts itself to the motion of the cardiac base in the heart cycle.3
As with any medical device implanted in a patient, it is crucial to ensure that the device successfully passes a stringent set of standards. To improve these standards, Falahatpisheh employs DPIV and computational fluid dynamics (CFD) to assess the flow passing through the valve. Using these techniques, he is able to determine how closely the artificial valves mimic the natural valve and ensure they are compliant with the standard guidelines. At this stage, it is essential to have a proper vortex pattern within the flow through the valve. Otherwise, the flow pattern is not optimized to properly transfer the momentum and energy from the heart chamber to the aorta.
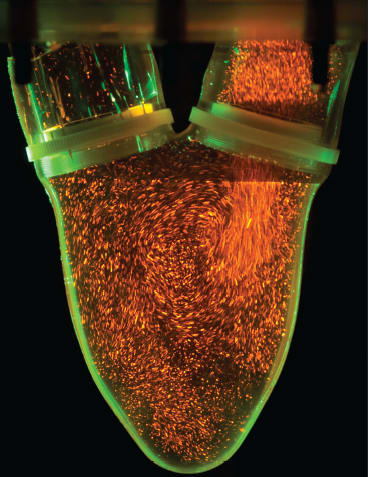
DPIV research on heart valves assures physicians and the Food and Drug Administration (FDA) that the heart valves implanted in a patient will operate safely and correctly. To conduct his DPIV studies, Falahatpisheh illuminates fluorescent micro particles on the plane from which he is interested in gathering data. From there, he takes 1,000 images per second to visualize the flow field. Previously, scientists could only capture 15 frames per second. Using visualization software, he imports the DPIV data related to the flow through a valve and identifies potential anomalies. Based on his results, Falahatpisheh can determine how and where to alter the design of the bileaflet mitral BHV to eliminate any anomalies and ensure a proper flow pattern. If this portion is inaccurate, all the work and research that takes place after the tests will also be inaccurate.
By using DPIV and the artificial heart flow simulator at KLAB, any pulsatile flow similar to the blood flow inside the heart can be experimentally reproduced, eliminating the difficulty of implementing boundary conditions for the CFD.
The Future of Artificial Valves
Technological advances will continue to facilitate the medical industry’s ability to make a greater impact on patients’ quality of life.
Falahatpisheh’s research to detect vortex abnormalities will further revolutionize these advancements, particularly by designing a dynamic and bileaflet mitral BHV that could last the patient’s lifetime.
This article was written by Scott Imlay, PhD, CTO, Tecplot, Inc., Bellevue, WA, and Ahmad Falahatpisheh, a Ph.D. candidate in the lab of Prof. Kheradvar (KLAB) at the University of California, Irvine. For more information, Click Here .
References
- Medscape Today
- The Society of Thoracic Surgeons
- Kheradvar A. and Falahatpisheh A., The Effects of Dynamic Saddle Annulus and Leaflet Length on Trasmitral Flow Pattern and Leaflet Stress of a Bi-leaflet Bioprosthetic Mitral Valve, J of Heart Valve Disease (Accepted)



